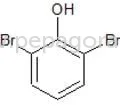
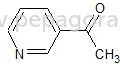
𝗪𝗵𝗮𝘁 𝗮𝗿𝗲 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹 𝗥𝗮𝘄 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀?
Pharmaceutical raw materials are the fundamental components used in the manufacturing of medications. These materials can be active pharmaceutical ingredients (APIs) or excipients that serve various functions, such as aiding delivery or enhancing stability. They are critical to the pharmaceutical industry as they form the basis of drug formulations and are essential in ensuring the efficacy and safety of medications.
These raw materials are utilized in several business applications, including drug development, production, and quality control. Pharmaceutical companies rely on high-quality raw materials to comply with regulatory standards and to deliver effective healthcare solutions. The industry is highly regulated, and companies must adhere to strict guidelines to ensure product quality and patient safety. The demand for these materials continues to grow, driven by advancements in drug development and an increase in global healthcare needs.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharmaceutical raw materials possess specific features that ensure their effectiveness in drug formulation. Understanding these specifications is crucial for manufacturers.
Key specifications include:
1. Active Pharmaceutical Ingredients (APIs)
- These ingredients are biologically active compounds that provide pharmacological activity.
- Specifications often include purity levels, solubility, and chemical stability.
2. Excipients
- Inactive substances that assist in the drug formulation process.
- Specifications may involve compatibility with APIs, moisture content, and particle size.
3. Quality Assurance
- Ensures that raw materials meet regulatory standards.
- Specifications can include testing methods and validation processes.
4. Storage Conditions
- Guidelines for temperature and humidity control.
- Specifications often include acceptable ranges for storage to maintain material integrity.
5. Packaging Requirements
- Standards for packaging materials to prevent contamination.
- Specifications may detail the types of materials used and their compatibility with the product.
6. Compliance Standards
- Adherence to industry regulations such as GMP (Good Manufacturing Practices).
- Specifications include documentation and certification requirements.
7. Shelf Life
- The duration for which the raw material remains effective and safe to use.
- Specifications often detail the stability studies conducted.
8. Testing Protocols
- Required tests to ensure material quality and safety.
- Specifications can include methods for testing purity, potency, and contaminants.
In summary, understanding the key features and specifications of pharmaceutical raw materials is essential for manufacturers to ensure product quality and compliance with regulatory standards.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Pharmaceutical raw materials serve a wide range of industries and applications.
1. Pharmaceutical Manufacturing: These materials are crucial in the production of medications, ensuring the correct formulation and dosage of drugs.
2. Biotechnology: Raw materials are used in the development of biologics and biosimilars, which require precise formulations and quality control.
3. Nutraceuticals: Pharmaceutical raw materials are integral in producing dietary supplements and functional foods that support health and wellness.
4. Cosmetics: Many pharmaceutical raw materials are used in the formulation of skincare products, ensuring safety and efficacy.
5. Veterinary Medicine: Raw materials are also applied in the development of medications for animals, ensuring effective treatments.
6. Research and Development: Pharmaceutical companies rely on these materials for clinical trials and studies aimed at discovering new drugs.
7. Contract Manufacturing: Many companies outsource the production of pharmaceutical products, which involves the use of quality raw materials.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
Pharmaceutical raw materials encompass a diverse range of products that cater to various needs.
Active Pharmaceutical Ingredients (APIs): These are the core components that provide therapeutic effects in medications. They vary in complexity and are vital for drug efficacy.
Excipients: These materials support the drug formulation process but do not have therapeutic effects. Variants include binders, fillers, and preservatives, each chosen for specific applications.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Sourcing high-quality pharmaceutical raw materials offers numerous advantages to manufacturers.
1. Quality Assurance: Ensures high standards of safety and efficacy in drug formulations, leading to better patient outcomes.
2. Regulatory Compliance: Helps manufacturers meet stringent industry regulations, reducing the risk of penalties and recalls.
3. Cost Efficiency: High-quality raw materials can improve production efficiency and reduce waste, lowering overall costs.
4. Enhanced Formulation Stability: Quality materials contribute to longer shelf life and greater stability in finished products.
5. Innovation Support: Access to diverse raw materials facilitates research and development of new formulations and therapies.
6. Improved Supply Chain Reliability: Reliable sourcing of raw materials ensures timely production and delivery of medications.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing pharmaceutical raw materials, several factors should be considered to ensure quality and compliance.
1. Quality Assessment: Evaluate the raw materials against established quality criteria, including purity and potency. Testing certifications and documentation should be requested.
2. Supplier Verification: Vet suppliers to ensure they adhere to industry standards and have a track record of reliability. This includes checking certifications and customer references.
3. Pricing and MOQ: Assess the cost of raw materials in relation to their quality. Minimum order quantities should be considered to align with production needs and budgets.
4. Delivery and Lead Times: Understand the logistics involved in sourcing raw materials. Reliable delivery timelines are crucial for maintaining production schedules.
5. Compliance Requirements: Ensure that the raw materials meet all regulatory standards specific to the industry and region. This includes understanding documentation and certification needs.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharmaceutical raw materials must adhere to various compliance and certification standards to ensure product safety and quality.
ISO 9001 is a widely recognized quality management standard that outlines requirements for consistent quality and improvement in processes. Companies that comply with ISO 9001 demonstrate a commitment to quality and customer satisfaction.
HACCP (Hazard Analysis and Critical Control Points) is essential for ensuring food safety and is also relevant in pharmaceutical manufacturing when raw materials are used in nutraceuticals.
Good Distribution Practice (GDP) ensures that the quality and integrity of pharmaceutical products are maintained throughout the supply chain. Compliance with GDP is critical for companies handling raw materials.
Certifications such as CE mark indicate compliance with European safety standards, which can enhance market access and consumer confidence.
Additionally, safety and testing requirements are paramount. They ensure that raw materials are free from contaminants and meet the necessary health and safety standards. Regional compliance considerations vary, and manufacturers must stay informed about local regulations.
𝗪𝗵𝘆 𝗦𝗼𝘂𝗿𝗰𝗲 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹 𝗥𝗮𝘄 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀 𝗳𝗿𝗼𝗺 𝗣𝗲𝗽𝗮𝗴𝗼𝗿𝗮?
Pepagora provides a trusted platform for sourcing pharmaceutical raw materials, offering several key advantages for B2B buyers.
Verified Supplier Network: Pepagora has established a thorough vetting process for suppliers, ensuring that only those who meet strict quality and compliance standards are included. This reduces risks associated with sourcing from unverified suppliers.
Streamlined RFQ Process: The quotation system at Pepagora simplifies the procurement process. Buyers can easily request quotes and receive competitive pricing from a variety of suppliers, saving time and effort.
Trade Assurance and Support: Pepagora offers buyer protection, ensuring that transactions are secure and that buyers receive the quality materials they expect. This support fosters trust and confidence in the purchasing process.
Regional Coverage: With a wide geographic reach, including India, the GCC, and Southeast Asia, Pepagora connects buyers with suppliers across multiple regions. This enhances sourcing options and allows for better negotiation of terms and pricing.
By choosing Pepagora, B2B buyers can confidently navigate the complexities of sourcing pharmaceutical raw materials while ensuring quality, compliance, and efficiency.