𝗪𝗵𝗮𝘁 𝗮𝗿𝗲 𝗔𝗻𝘁𝗶𝗯𝗶𝗼𝘁𝗶𝗰 𝗥𝗮𝘄 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀?
Antibiotic raw materials are the fundamental components used in the production of antibiotics, which are critical in the fight against bacterial infections. These raw materials comprise active pharmaceutical ingredients (APIs) and excipients that ensure the efficacy, safety, and stability of antibiotic formulations. They are manufactured through various chemical processes or extracted from natural sources, and they serve as the building blocks for pharmaceutical companies developing antibiotics for therapeutic use.
The primary applications of antibiotic raw materials are in the pharmaceutical industry, where they are used to create medications that combat bacterial infections in humans and animals. These materials are essential for the formulation of different antibiotic classes, including penicillins, cephalosporins, and macrolides. Additionally, they play a vital role in veterinary medicine for treating infections in livestock and pets. The growing global demand for antibiotics, driven by rising antibiotic resistance and the need for new treatments, highlights the importance of these raw materials in the healthcare supply chain.
In the current market context, the antibiotic raw materials industry is highly competitive and regulated. Manufacturers must adhere to stringent quality standards and regulations to ensure the safety and efficacy of their products. As antibiotic resistance continues to be a pressing public health challenge, the demand for innovative antibiotic formulations and their corresponding raw materials is expected to increase, driving growth in this sector.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Antibiotic raw materials are designed with specific features and specifications that ensure their effectiveness and safety in pharmaceutical applications. These specifications help manufacturers maintain quality and compliance with regulatory standards.
Key specifications include:
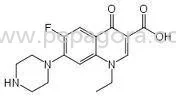
1. Active Pharmaceutical Ingredients (APIs)
- The primary component that provides therapeutic effects.
- Quality and purity must meet pharmacopoeial standards.
2. Excipients
- Inactive substances that aid in drug formulation.
- Must be non-toxic and compatible with active ingredients.
3. Solubility
- The ability of the antibiotic to dissolve in solvents.
- Important for bioavailability and effectiveness.
4. Stability
- The ability of the antibiotic to maintain its properties over time.
- Stability testing is crucial to ensure shelf life.
5. Particle Size Distribution
- Affects the solubility and bioavailability of the antibiotic.
- Must be controlled within specified limits.
6. Moisture Content
- Should be minimized to prevent degradation.
- Typically specified in percentage ranges.
7. pH Levels
- The acidity or alkalinity of the raw material.
- Must be within a specified range for stability.
8. Regulatory Compliance
- Must meet industry standards such as CE certification.
- Compliance with GMP (Good Manufacturing Practice) is essential.
In summary, the features and specifications of antibiotic raw materials are critical for ensuring quality and effectiveness in pharmaceutical applications, making them vital for manufacturers in the industry.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Antibiotic raw materials serve various industries, primarily focused on healthcare and pharmaceuticals.
1. Pharmaceutical Industry: These raw materials are essential in the production of oral and injectable antibiotics, enabling the treatment of a range of bacterial infections in humans.
2. Veterinary Medicine: Antibiotic raw materials are used in formulating medications for livestock and pets, ensuring animal health and reducing disease spread.
3. Biotechnology: In biopharmaceutical development, these materials are crucial for research and production of novel antibiotics to combat resistant strains of bacteria.
4. Agriculture: Antibiotic products derived from these raw materials are employed to promote growth and prevent diseases in livestock.
5. Research Institutions: Laboratories utilize antibiotic raw materials for developing new drugs and studying bacterial resistance mechanisms.
6. Compounding Pharmacies: These facilities use raw materials to create customized antibiotic formulations tailored to patient needs.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
Antibiotic raw materials are diverse, catering to various pharmaceutical needs and applications.
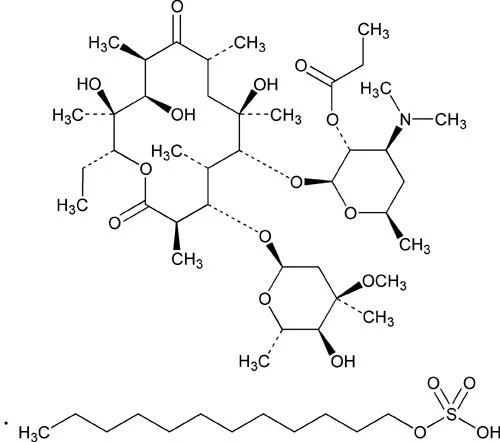
Penicillin Derivatives
These variants include various forms of penicillin, which are widely used to treat infections caused by susceptible bacteria. They are characterized by their effectiveness against gram-positive bacteria and are fundamental in many antibiotic formulations.
Cephalosporin Compounds
This category includes a range of cephalosporins that provide broad-spectrum antibacterial activity. They are often used in surgical prophylaxis and to treat serious infections.
Macrolide Antibiotics
These raw materials are used to produce macrolide antibiotics, which are effective against a variety of bacterial infections. They are particularly useful for patients allergic to penicillin.
Tetracycline Compounds
This variant includes tetracyclines that are used in both human and veterinary medicine. They are known for their broad-spectrum activity against numerous bacteria.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Sourcing antibiotic raw materials offers numerous benefits to pharmaceutical companies.
1. Quality Assurance: High-quality raw materials ensure the effectiveness and safety of final antibiotic products, reducing the risk of treatment failures.
2. Regulatory Compliance: Sourcing from certified suppliers helps companies meet stringent regulatory requirements, ensuring smooth market entry.
3. Cost Efficiency: Competitive pricing from reliable suppliers can lower manufacturing costs while maintaining quality standards.
4. Customization: Suppliers may offer tailored formulations to meet specific therapeutic needs, enhancing product versatility.
5. Supply Chain Reliability: A stable supply of raw materials minimizes disruptions in production and ensures timely delivery of finished products.
6. Innovation Support: Access to advanced raw materials can facilitate the development of new antibiotics, addressing emerging resistance challenges.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing antibiotic raw materials, various factors must be considered to ensure a successful procurement process.
1. Quality Assessment: Evaluate the quality of raw materials by reviewing certificates of analysis and compliance with pharmacopoeial standards. Thorough testing for purity and potency is essential.
2. Supplier Verification: Conduct thorough background checks on suppliers, including their manufacturing practices and certifications. A reliable supplier should have a proven track record in the industry.
3. Pricing and MOQ: Understand the pricing structure and minimum order quantities (MOQ) offered by suppliers. This helps in budgeting and ensures that the procurement aligns with production needs.
4. Delivery and Lead Times: Discuss logistics with suppliers to establish realistic delivery schedules. Timely delivery is crucial for maintaining production timelines.
5. Compliance Requirements: Ensure that the raw materials meet all relevant regulatory and safety standards. This includes certifications such as CE and adherence to GMP guidelines.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Antibiotic raw materials must comply with various quality standards and regulations to ensure safety and efficacy in pharmaceutical applications. Key standards include ISO 9001, which focuses on quality management systems, and HACCP, which emphasizes food safety. Compliance with GDP (Good Distribution Practice) is also essential to ensure that products are stored and transported under appropriate conditions.
Industry certifications, such as CE marking, indicate that the products meet European safety and health requirements. Additionally, raw materials must undergo rigorous safety and testing requirements to confirm their stability and compatibility in antibiotic formulations.
Regional compliance considerations may vary, with specific regulations governing the production and distribution of pharmaceutical ingredients in different countries. It's crucial for manufacturers to be aware of these regulations to ensure adherence and avoid potential legal issues.
𝗪𝗵𝘆 𝗦𝗼𝘂𝗿𝗰𝗲 𝗔𝗻𝘁𝗶𝗯𝗶𝗼𝘁𝗶𝗰 𝗥𝗮𝘄 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀 𝗳𝗿𝗼𝗺 𝗣𝗲𝗽𝗮𝗴𝗼𝗿𝗮?
Pepagora provides a trusted platform for sourcing antibiotic raw materials, offering several key advantages to B2B buyers.
Verified Supplier Network: Pepagora has established a robust network of verified suppliers who adhere to stringent quality standards. Through rigorous vetting processes, buyers can ensure they are working with reputable manufacturers.
Streamlined RFQ Process: The Request for Quotation (RFQ) process on Pepagora is designed for efficiency, allowing buyers to receive multiple quotations from suppliers quickly. This simplifies the procurement process and helps in making informed purchasing decisions.
Trade Assurance and Support: Pepagora offers trade assurance to protect buyers against potential risks, ensuring that transactions are secure and that suppliers deliver as promised. This support fosters trust and confidence in the sourcing process.
Regional Coverage: With a focus on markets in India, the GCC, and Southeast Asia, Pepagora provides extensive regional coverage. This enables buyers to access a diverse range of antibiotic raw materials tailored to their specific needs, while also considering logistical aspects of procurement.