𝗪𝗵𝗮𝘁 𝗮𝗿𝗲 𝗣𝗵𝗮𝗿𝗺𝗮 𝗣𝗮𝗰𝗸𝗮𝗴𝗶𝗻𝗴 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀?
Pharma packaging materials are specialized products designed to protect pharmaceuticals during storage and transportation. These materials are essential for maintaining the integrity, safety, and efficacy of medications. They can include various types of packaging, such as containers, labels, and closures, made from materials like glass, plastic, and metal. Each type of packaging is engineered to meet specific requirements related to the pharmaceutical products they contain.
The primary uses of pharma packaging materials extend beyond mere containment. They play a critical role in ensuring compliance with regulatory standards, preventing contamination, and extending shelf life. These materials are increasingly utilized in both bulk and retail packaging, serving manufacturers, wholesalers, and pharmacies. The pharmaceutical industry demands high-quality packaging solutions that are not only functional but also capable of conveying vital information to consumers and healthcare providers.
In terms of market context, the pharma packaging sector is growing rapidly due to increased demand for pharmaceuticals worldwide. This growth is driven by advancements in drug development and an aging population requiring more medications. Consequently, the need for innovative and sustainable packaging solutions is becoming more pronounced, making pharma packaging materials a vital part of the pharmaceutical supply chain.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharma packaging materials must meet stringent performance criteria and specifications to ensure they effectively protect the products. These specifications often vary based on the type of pharmaceutical product and its intended use.
Key specifications include:
1. Material Type
- Common materials include glass, polyethylene, polypropylene, and aluminum.
- Each material has unique properties such as barrier protection and chemical compatibility.
2. Barrier Properties
- Essential for preventing moisture, oxygen, and light from degrading the product.
- Specific barrier levels are often tested and reported in compliance with industry standards.
3. Compliance Standards
- Must adhere to regulations such as Good Manufacturing Practices (GMP) and ISO standards.
- Certifications like CE indicate compliance with health, safety, and environmental protection standards.
4. Customization Options
- Packaging can be tailored to meet specific branding and functional requirements.
- Custom sizes, shapes, and printing are available based on client needs.
5. Sustainability Features
- Increasing emphasis on eco-friendly materials and recyclable packaging options.
- Many manufacturers are moving towards biodegradable alternatives.
6. Temperature Stability
- Packaging must maintain stability under various temperature conditions.
- Specifications often include ranges suitable for different pharmaceutical products.
7. Shelf Life and Storage Conditions
- Packaging must provide adequate protection to extend the shelf life of the medication.
- Specifications often include recommended storage conditions to maintain product integrity.
In summary, the key features and specifications of pharma packaging materials are critical to ensuring product safety, compliance, and effectiveness. Buyers must carefully evaluate these aspects when selecting packaging solutions.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Pharma packaging materials are utilized across various industries, each with specific applications.
1. Pharmaceutical Manufacturers: These companies require packaging that can protect drugs during production, storage, and distribution. The materials used must comply with strict regulations to ensure safety and efficacy.
2. Biotech Firms: Biotech products often require specialized packaging to maintain stability and protect sensitive formulations. These materials must provide robust barrier properties.
3. Over-the-Counter Products: Packaging for OTC medications must be user-friendly and include clear labeling. Consumer safety is a top priority in this segment.
4. Veterinary Medicine: Packaging for veterinary pharmaceuticals must be robust enough to handle the specific needs of animal health products, including dosage forms and storage conditions.
5. Nutraceuticals: As the nutraceutical market grows, packaging solutions must address the unique requirements of dietary supplements, including child-resistant features and moisture barriers.
6. Clinical Trials: Packaging used in clinical trials must ensure product integrity while being easy to identify and use for participants. It often includes blinding and labeling considerations.
7. Home Healthcare: With the rise of home healthcare, packaging must be designed for convenience and safety, ensuring that patients can easily access their medications.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
Pharma packaging materials come in various forms to meet specific needs of different products.
Glass Containers:
Glass containers are commonly used for pharmaceuticals due to their excellent barrier properties and inertness. They are ideal for liquid medications, especially those sensitive to light and moisture.
Plastic Bottles:
Plastic bottles are lightweight and shatter-resistant, making them suitable for both solid and liquid pharmaceuticals. They often come with child-resistant closures for safety.
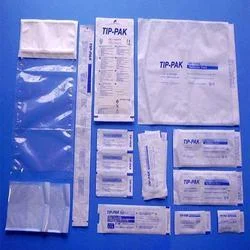
Blister Packs:
Blister packs provide unit-dose packaging for tablets and capsules, ensuring product protection and ease of use. They are designed to maintain product integrity and are often used in both retail and clinical settings.
Syringes and Vials:
These are essential for injectable medications. They are designed for ease of use and must meet stringent sterility requirements.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Pharma packaging materials offer numerous benefits that are essential for product integrity and compliance.
1. Enhanced Product Safety: Proper packaging protects medications from contamination and degradation, ensuring they remain safe for consumption.
2. Regulatory Compliance: Using compliant packaging helps manufacturers meet industry regulations, reducing the risk of legal issues.
3. Improved Shelf Life: High-quality materials extend the shelf life of pharmaceuticals, which is crucial for maintaining efficacy and reducing waste.
4. User Convenience: Packaging designed with user experience in mind makes it easier for consumers to access their medications correctly.
5. Brand Differentiation: Custom packaging solutions allow companies to enhance their brand image and stand out in a competitive market.
6. Environmental Sustainability: Many manufacturers are now offering eco-friendly packaging options, appealing to environmentally conscious consumers.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing pharma packaging materials, B2B buyers should consider several key factors to ensure they make informed decisions.
1. Quality Assessment: Evaluate the quality of packaging materials based on performance standards and testing results. Look for certifications that indicate compliance with industry regulations.
2. Supplier Verification: Vet suppliers to ensure they have a track record of providing reliable, high-quality products. Check for customer reviews and industry certifications to establish credibility.
3. Pricing and MOQ: Understand the pricing structure and minimum order quantities (MOQ) required by suppliers. This information is crucial for budgeting and inventory management.
4. Delivery and Lead Times: Assess the supplier's ability to meet delivery schedules. Timely delivery is essential to maintain production timelines.
5. Compliance Requirements: Ensure that the selected packaging materials meet relevant regulatory requirements, including local and international standards.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharma packaging materials must comply with various quality standards to ensure safety and effectiveness.
Relevant quality standards include ISO 9001, which addresses quality management systems, and HACCP, which focuses on food safety management. Additionally, Good Distribution Practice (GDP) guidelines are critical for ensuring that pharmaceutical products are consistently stored, transported, and distributed under suitable conditions.
Industry certifications, such as CE, signify that products meet health, safety, and environmental protection standards. This certification is particularly important in Europe and can influence market entry and acceptance.
Safety and testing requirements must also be met, as packaging materials must be tested for factors like chemical compatibility, sterility, and material integrity.
Regional compliance considerations vary depending on the market, and buyers should be aware of local regulations that may impact packaging requirements.
𝗪𝗵𝘆 𝗦𝗼𝘂𝗿𝗰𝗲 𝗣𝗵𝗮𝗿𝗺𝗮 𝗣𝗮𝗰𝗸𝗮𝗴𝗶𝗻𝗴 𝗠𝗮𝘁𝗲𝗿𝗶𝗮𝗹𝘀 𝗳𝗿𝗼𝗺 𝗣𝗲𝗽𝗮𝗴𝗼𝗿𝗮?
Pepagora provides a trusted platform for sourcing pharma packaging materials, offering several key advantages to B2B buyers.
Verified Supplier Network: Pepagora has established a rigorous vetting process to ensure that all suppliers meet high standards of quality and reliability. This helps buyers find trustworthy partners quickly.
Streamlined RFQ Process: The platform simplifies the request for quotation (RFQ) process, allowing buyers to easily compare options and make informed purchasing decisions. This efficiency saves time and resources.
Trade Assurance and Support: Pepagora offers trade assurance to protect buyers, ensuring that they receive products that meet their specifications. Dedicated customer support is also available to assist with any inquiries.
Regional Coverage: With a strong presence in India, the GCC, and Southeast Asia, Pepagora connects buyers with suppliers across these regions, facilitating access to a diverse range of pharma packaging materials. This geographic reach enhances sourcing options for businesses looking to expand their supply chains.