What are Active Pharmaceutical Ingredients (APIs)?
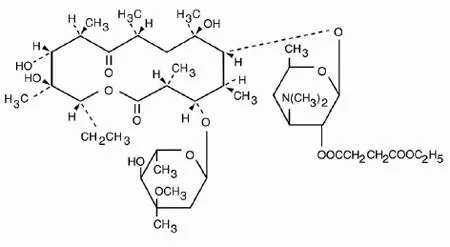
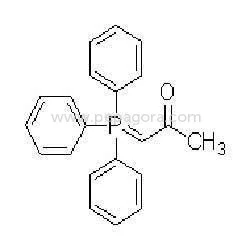
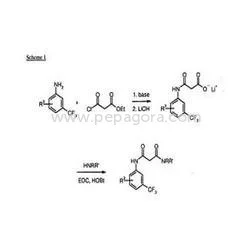
Active Pharmaceutical Ingredients, or APIs, are the active components in pharmaceutical drugs that provide therapeutic effects. APIs can be derived from natural sources or synthesized chemically. They are essential for the efficacy of medications, making them a critical focus for pharmaceutical manufacturers. Each API has specific pharmacological properties that determine how it interacts within the body.
APIs are used in various applications, including the production of prescription medications, over-the-counter drugs, and biologics. They serve a vital role in the pharmaceutical industry by ensuring that medications are effective, safe, and reliable. The market for APIs is expanding, driven by the growing demand for innovative treatments and personalized medicine. Companies involved in drug development rely heavily on high-quality APIs to maintain their competitive edge and meet regulatory standards.
The relevance of APIs extends beyond individual medications; they are integral to the broader healthcare landscape. With the rise of generic drugs and the increasing complexity of drug formulations, the API market is continuously evolving. This dynamic environment presents both challenges and opportunities for manufacturers, suppliers, and end-users alike.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Active Pharmaceutical Ingredients are characterized by several key features that ensure their effectiveness and compliance with industry standards. Understanding these specifications can aid businesses in selecting the right APIs for their formulations.
Key specifications include:
1. Purity Level
- High purity is essential for ensuring safety and efficacy.
- Typically, APIs have a purity level of 98% or higher.
2. Solubility
- The ability of an API to dissolve in solvents affects its bioavailability.
- Solubility parameters vary widely; some APIs are highly soluble while others are not.
3. Stability
- APIs must maintain their effectiveness over time and under various conditions.
- Stability is often measured in terms of shelf life and reaction to environmental factors.
4. Particle Size Distribution
- The size of API particles can impact drug absorption and bioavailability.
- Manufacturers often specify a range for optimal performance.
5. Moisture Content
- Excess moisture can lead to degradation of APIs.
- Specifications may dictate moisture content to be below a certain percentage.
6. Regulatory Compliance
- APIs must adhere to various regulatory standards, such as Good Manufacturing Practice (GMP).
- Compliance is critical for market acceptance and safety assurance.
7. Formulation Compatibility
- APIs should be compatible with other excipients used in drug formulations.
- Compatibility testing is essential for ensuring product integrity.
8. Source and Origin
- The origin of an API can affect its regulatory status and quality assurance.
- APIs can be sourced from synthetic processes or natural extractions.
Overall, understanding these features and specifications is crucial for pharmaceutical companies aiming to develop effective and safe medications.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Active Pharmaceutical Ingredients are utilized across several industries, highlighting their versatility and importance.
1. Pharmaceuticals: APIs are the cornerstone of drug development, providing the active components in both branded and generic medications.
2. Biotechnology: In biologics, APIs derived from living organisms are crucial in treatments for various diseases, including cancers and autoimmune disorders.
3. Veterinary Medicine: APIs are used to formulate medications for animals, ensuring they receive effective treatments for various ailments.
4. Nutraceuticals: Some APIs are incorporated into dietary supplements, enhancing health benefits and therapeutic claims.
5. Cosmetic Products: Certain APIs are used in cosmetic formulations to provide therapeutic effects, such as anti-aging properties.
6. Research and Development: APIs are essential in clinical trials, providing the basis for testing new drugs and therapies.
7. Personal Care: APIs are increasingly being integrated into personal care products for their beneficial health effects.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
Active Pharmaceutical Ingredients come in various forms, each tailored for specific uses and applications. This diversity allows pharmaceutical companies to select the most suitable API for their products.
Variant 1: Injectable APIs
Injectable APIs are specifically designed for formulations intended for parenteral administration. They often require strict sterility and stability standards, making them crucial for effective treatments in hospitals and clinics.
Variant 2: Oral APIs
Oral APIs are formulated for tablets and capsules. They must be optimized for absorption and bioavailability, ensuring they deliver the intended therapeutic effect when ingested.
Variant 3: Topical APIs
Topical APIs are used in creams, ointments, and gels, designed for local application on the skin. These APIs focus on delivering localized treatment while minimizing systemic absorption.
Variant 4: Inhalation APIs
Inhalation APIs are specially formulated for respiratory therapies, providing quick relief from conditions like asthma and COPD. They must be compatible with delivery devices to ensure effectiveness.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Sourcing high-quality Active Pharmaceutical Ingredients provides numerous advantages for pharmaceutical companies.
1. Enhanced Efficacy: High-quality APIs ensure that medications perform as intended, leading to better patient outcomes.
2. Regulatory Compliance: Reliable APIs adhere to strict regulatory standards, reducing the risk of compliance-related issues.
3. Cost-Effectiveness: Sourcing APIs from reputable suppliers can lead to cost savings through bulk purchases and reduced waste.
4. Reliability: Consistent quality and supply chain stability are critical, and established suppliers often provide both.
5. Innovation Opportunities: Access to diverse APIs allows companies to innovate and develop new formulations tailored to market needs.
6. Competitive Advantage: High-quality APIs can differentiate products in a crowded market, attracting more customers.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing Active Pharmaceutical Ingredients, B2B buyers should consider several critical factors to ensure they obtain the best products.
1. Quality Assessment: Evaluate the quality of APIs by reviewing purity levels, stability data, and compliance with industry standards. This may involve requesting Certificates of Analysis from suppliers.
2. Supplier Verification: Conduct thorough background checks on potential suppliers. Look for certifications, customer reviews, and industry reputation to ensure reliability.
3. Pricing and MOQ: Understand the pricing structures and minimum order quantities (MOQ) that suppliers offer. This information is crucial for budgeting and inventory management.
4. Delivery and Lead Times: Clarify delivery schedules and lead times to ensure timely access to APIs. Delays can impact production schedules and market readiness.
5. Compliance Requirements: Verify that suppliers meet all relevant regulatory and compliance standards specific to the target market. This includes understanding local regulations and certifications.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Active Pharmaceutical Ingredients must adhere to various quality standards to ensure safety and efficacy. Key standards include ISO 9001, which focuses on quality management systems, and Good Distribution Practice (GDP), which ensures the integrity of the supply chain.
Industry certifications, such as CE marking, indicate compliance with safety and health requirements in the European market. Compliance with these standards helps mitigate risks associated with drug safety.
Safety and testing requirements are also integral to the API lifecycle. Rigorous testing for contaminants, stability, and efficacy must be conducted to meet regulatory expectations.
Regional compliance considerations can vary significantly. Different countries may impose unique requirements for APIs, making it essential for suppliers to stay informed about local regulations and standards.
Why Source Active Pharmaceutical Ingredients (APIs) from Pepagora?
Pepagora provides an efficient and trusted platform for sourcing Active Pharmaceutical Ingredients, catering to the diverse needs of the pharmaceutical industry.
Verified Supplier Network: Pepagora’s network consists of thoroughly vetted suppliers, ensuring that buyers can source high-quality APIs with confidence. This verification process reduces the risks associated with procurement.
Streamlined RFQ Process: The Request for Quotation (RFQ) system simplifies the procurement process. Buyers can easily compare offers, negotiate terms, and select the best suppliers for their needs.
Trade Assurance and Support: Pepagora offers trade assurance to protect buyers throughout their purchasing journey. This support includes assistance with logistics, quality assurance, and dispute resolution.
Regional Coverage: With a strong presence in India, the GCC, and Southeast Asia, Pepagora provides extensive geographic reach. This allows buyers to access a wide range of APIs from trusted sources across these regions, enhancing their supply chain flexibility.
By choosing Pepagora, businesses can navigate the complexities of sourcing Active Pharmaceutical Ingredients efficiently and effectively, ensuring they obtain the best products for their pharmaceutical applications.