𝗪𝗵𝗮𝘁 𝗮𝗿𝗲 𝗣𝗿𝗲𝘀𝗲𝗿𝘃𝗮𝘁𝗶𝘃𝗲𝘀 𝗳𝗼𝗿 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹𝘀?
Preservatives for pharmaceuticals are substances added to drug formulations to prevent microbial growth and spoilage. These chemicals help maintain the stability and efficacy of pharmaceutical products, ensuring they remain safe and effective for consumer use. Preservatives are essential components, particularly in liquid formulations, where the risk of contamination is higher. They play a crucial role in extending the shelf life of products and protecting them from degradation caused by environmental factors.
In the pharmaceutical industry, preservatives are used in various applications, including injectable solutions, topical creams, and oral medications. Their primary purpose is to inhibit the growth of bacteria, fungi, and other microorganisms that could compromise the integrity of the product. The relevance of preservatives in pharmaceuticals is underscored by stringent regulatory requirements and industry standards aimed at ensuring patient safety. As the market continues to evolve with increasing demand for safe and effective medications, the importance of selecting the right preservatives has never been more critical.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Preservatives for pharmaceuticals offer a range of features designed to meet industry standards and ensure product efficacy. These specifications are essential for manufacturers looking to maintain compliance and enhance the quality of their products.
Key specifications include:
1. Efficacy Spectrum
- Ability to inhibit a wide range of microorganisms
- Includes bacteria, yeast, and mold
2. Chemical Stability
- Resistance to degradation under various conditions
- Maintains effectiveness over the product's shelf life
3. Compatibility
- Safe for use in various pharmaceutical formulations
- Does not negatively affect the product's active ingredients
4. Regulatory Compliance
- Meets industry standards such as CE certification
- Adheres to local and international regulations
5. Concentration Levels
- Recommended dosage levels vary based on formulation type
- Ensures optimal performance without compromising safety
6. pH Range
- Effective across a wide range of pH levels
- Supports stability in acidic or alkaline formulations
7. Solubility
- Easily soluble in water or other solvents used in formulations
- Ensures uniform distribution within the product
8. Toxicity Profile
- Low toxicity to ensure safety for end-users
- Evaluated through thorough safety assessments
In summary, the specifications of preservatives for pharmaceuticals are vital for ensuring product effectiveness, safety, and compliance with regulatory standards.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Preservatives for pharmaceuticals are utilized across various industries to enhance product longevity and safety.
1. Pharmaceutical Industry: Preservatives are commonly used in injectable solutions to prevent microbial contamination, ensuring patient safety and product integrity.
2. Cosmetic Industry: In topical creams and lotions, preservatives prevent spoilage and extend shelf life, allowing products to remain effective for longer periods.
3. Nutraceuticals: Preservatives are added to dietary supplements to maintain potency and prevent degradation, particularly in liquid formulations.
4. Veterinary Medicine: Preservatives are used in veterinary pharmaceuticals to ensure the safety and effectiveness of medications for animals.
5. Biologics: In the production of biologics, preservatives help maintain the stability of sensitive formulations, which are crucial for patient treatment.
6. Diagnostics: Preservatives are essential in diagnostic reagents to prevent microbial growth and ensure accurate results.
7. Personal Care Products: In personal care formulations, preservatives help extend the product's usability while maintaining safety for consumers.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
The diversity of preservatives for pharmaceuticals allows manufacturers to choose the most suitable option for their specific formulations.
Variant 1 Name: Benzyl Alcohol
Benzyl alcohol is commonly used as a preservative in injectable medications. It has antimicrobial properties and is effective in preventing the growth of bacteria and fungi. This variant is favored for its compatibility with various active ingredients.
Variant 2 Name: Phenol
Phenol is another widely used preservative that offers broad-spectrum antimicrobial activity. It is particularly effective in topical formulations, providing protection against microbial contamination while enhancing the stability of the product.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Utilizing preservatives in pharmaceuticals offers numerous benefits that contribute to product quality and safety.
1. Enhanced Stability: Preservatives help maintain the chemical integrity of pharmaceutical products, extending shelf life and ensuring efficacy.
2. Microbial Protection: They prevent the growth of harmful microorganisms, safeguarding patients from potential infections or adverse reactions.
3. Cost-Effectiveness: By extending the usability of products, preservatives help reduce waste and lower production costs for manufacturers.
4. Compliance Assurance: Using preservatives that meet regulatory standards ensures compliance with industry requirements, reducing the risk of product recalls.
5. Versatility: A wide range of preservatives is available, allowing manufacturers to select options that best suit their specific formulations.
6. Improved Consumer Confidence: Products with proper preservation instill trust in consumers, ensuring they feel safe using the medications.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing preservatives for pharmaceuticals, it is essential for businesses to consider several critical factors to ensure they make informed decisions.
1. Quality Assessment: Evaluate the quality of preservatives based on their efficacy, stability, and compliance with industry standards. Look for certifications that indicate reliability.
2. Supplier Verification: Vet suppliers to ensure they have a proven track record in the pharmaceutical industry. Check references and certifications to guarantee product quality.
3. Pricing and MOQ: Understand the cost structure and minimum order quantities. Compare prices among different suppliers to find the best value without compromising quality.
4. Delivery and Lead Times: Inquire about logistics and delivery schedules. Timely delivery is crucial in maintaining production schedules and meeting market demands.
5. Compliance Requirements: Ensure that the preservatives meet local and international regulatory standards. This is essential to avoid legal issues and ensure patient safety.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
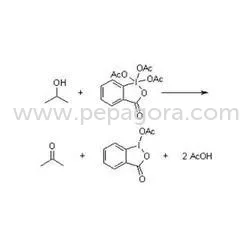
Preservatives for pharmaceuticals must adhere to several relevant quality standards and certifications to ensure safety and effectiveness. Compliance with ISO 9001 quality management systems is crucial, as it indicates that manufacturers meet international standards for quality assurance.
HACCP (Hazard Analysis and Critical Control Points) is another important standard that focuses on ensuring food safety and preventing contamination throughout the production process. Additionally, compliance with GDP (Good Distribution Practice) ensures that products are stored and transported under appropriate conditions to maintain their integrity.
Industry certifications, such as CE marking, indicate that a product meets the essential requirements of European health, safety, and environmental protection legislation. Furthermore, safety and testing requirements must be met to ensure that preservatives do not pose risks to consumers.
Regional compliance considerations vary, and companies must stay informed about local regulations to ensure their products meet all necessary standards for the markets they serve.
𝗪𝗵𝘆 𝗦𝗼𝘂𝗿𝗰𝗲 𝗣𝗿𝗲𝘀𝗲𝗿𝘃𝗮𝘁𝗶𝘃𝗲𝘀 𝗳𝗼𝗿 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹𝘀 𝗳𝗿𝗼𝗺 𝗣𝗲𝗽𝗮𝗴𝗼𝗿𝗮?
Pepagora offers a trusted platform for sourcing preservatives for pharmaceuticals, providing several key advantages to B2B buyers.
Verified Supplier Network: Our platform connects buyers with a network of verified suppliers who have undergone thorough vetting to ensure product quality and reliability. This minimizes risks and enhances trust between buyers and suppliers.
Streamlined RFQ Process: Pepagora features a user-friendly request for quotation (RFQ) system that simplifies the sourcing process. Buyers can easily submit requests, receive quotations, and compare offerings from multiple suppliers.
Trade Assurance and Support: We prioritize buyer protection through trade assurance measures, ensuring that transactions are secure and that buyers receive the products as expected. Our dedicated customer support team is available to assist buyers throughout the sourcing process.
Regional Coverage: Pepagora has a geographical reach across India, the GCC, and Southeast Asia, making it easier for businesses to connect with suppliers in these regions. This broad coverage ensures that buyers have access to a wide range of products and suppliers.