𝗪𝗵𝗮𝘁 𝗮𝗿𝗲 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹 𝗜𝗻𝘁𝗲𝗿𝗺𝗲𝗱𝗶𝗮𝘁𝗲𝘀?
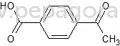
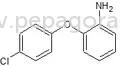
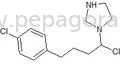
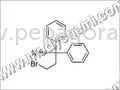
Pharmaceutical intermediates are chemical compounds that serve as precursors in the synthesis of active pharmaceutical ingredients (APIs). These intermediates are essential in the production process, helping transform raw materials into finished drugs. They play a critical role in the pharmaceutical industry by providing the necessary building blocks for a wide array of medicinal products.
These intermediates are primarily used in the development and manufacturing of pharmaceuticals, including generic drugs and over-the-counter medications. They are crucial for ensuring the efficacy and safety of the final products. Pharmaceutical intermediates are also utilized in research and development, allowing pharmaceutical companies to innovate and create new therapies. The relevance of pharmaceutical intermediates is growing as the demand for personalized medicine and complex drug formulations increases in the global market.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharmaceutical intermediates are characterized by several key features that determine their suitability for various applications. Understanding these specifications is vital for businesses seeking to procure high-quality intermediates for their production needs.
Key specifications include:
1. Purity Level
- Typically ranges from 95% to 99.9%
- Higher purity levels are essential for compliance with regulatory standards
2. Chemical Stability
- Must remain stable under specific temperature and humidity conditions
- Stability ensures the reliability of the final pharmaceutical product
3. Solubility
- Varies based on the specific intermediate
- Important for determining the formulation of the final product
4. Shelf Life
- Generally spans from one to three years
- Extended shelf life is critical for inventory management
5. Cost-Effectiveness
- Pricing must align with market standards to ensure competitiveness
- Bulk purchasing often reduces overall costs
6. Regulatory Compliance
- Must adhere to industry standards such as CE certification
- Compliance ensures safety and efficacy in pharmaceutical applications
In summary, selecting pharmaceutical intermediates with the right specifications is essential for ensuring product integrity and meeting regulatory requirements.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Pharmaceutical intermediates are utilized across various industries, reflecting their versatility and importance.
1. Pharmaceutical Industry: These intermediates are fundamental in the production of APIs for both prescription and over-the-counter medications, ensuring the safety and effectiveness of drugs.
2. Biotechnology: Pharmaceutical intermediates are used in the development of biologics, including monoclonal antibodies and vaccines, which require precise chemical processes.
3. Agrochemical Sector: Intermediates play a role in formulating agrochemicals, which are critical for enhancing crop yield and managing pests.
4. Nutraceuticals: These intermediates are involved in creating dietary supplements that require specific active ingredients for health benefits.
5. Cosmetic Products: Certain pharmaceutical intermediates are used in the formulation of skin care and beauty products, enhancing their effectiveness.
6. Research Institutions: Pharmaceutical intermediates are essential in laboratory settings for drug discovery and development, helping researchers create new therapies.
7. Veterinary Medicine: Intermediates are also used in the production of veterinary pharmaceuticals, ensuring the health and well-being of animals.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
The diversity of pharmaceutical intermediates allows for specific applications tailored to various needs in the industry.
Variant 1: Amino Acids
Amino acids serve as building blocks for proteins and are essential in the production of numerous pharmaceuticals. They are widely used in formulations for both human and animal health.
Variant 2: Alkaloids
Alkaloids are a class of chemical compounds that occur naturally in plants. They are used in the development of various medications, particularly pain management and anti-cancer drugs.
Variant 3: Peptides
Peptides are short chains of amino acids and are increasingly important in drug development due to their specificity and efficacy in targeting disease pathways.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Sourcing pharmaceutical intermediates offers several advantages that can enhance production efficiency and product quality.
1. Quality Assurance: High-quality intermediates ensure reliable outcomes in drug formulations, enhancing the overall safety of the final product.
2. Cost Savings: Bulk purchasing and strategic sourcing often lead to significant cost reductions, improving profitability for manufacturers.
3. Regulatory Compliance: Sourcing from certified suppliers ensures adherence to industry regulations, minimizing the risk of non-compliance issues.
4. Supply Chain Reliability: A diverse supplier network helps mitigate risks associated with supply chain disruptions, ensuring timely deliveries.
5. Innovation Support: Access to a range of intermediates allows companies to explore new formulations and innovative drug development.
6. Technical Support: Many suppliers offer expertise and support in selecting the right intermediates for specific applications.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing pharmaceutical intermediates, it is crucial to consider several factors to ensure optimal procurement decisions.
1. Quality Assessment: Evaluate the purity, stability, and consistency of intermediates. Look for suppliers who provide detailed certificates of analysis.
2. Supplier Verification: Vet potential suppliers by checking their certifications, compliance history, and industry reputation to ensure they meet your standards.
3. Pricing and MOQ: Understand the pricing structure and minimum order quantities required by suppliers. Compare offers from multiple vendors to ensure competitiveness.
4. Delivery and Lead Times: Confirm delivery schedules and lead times. Reliable logistics are essential for maintaining production timelines.
5. Compliance Requirements: Ensure that the intermediates meet all relevant regulatory standards and certifications, especially if your products are destined for highly regulated markets.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Compliance with industry standards and certifications is critical for pharmaceutical intermediates. Relevant quality standards such as ISO 9001 and Good Manufacturing Practice (GMP) ensure that intermediates are produced consistently and safely. Certification from organizations like CE indicates that the products meet specific safety and efficacy requirements.
Pharmaceutical intermediates must also undergo rigorous testing to ensure they are free from contaminants and meet purity specifications. Regional regulations may vary, requiring suppliers to adapt their processes to comply with local laws and market demands.
𝗪𝗵𝘆 𝗦𝗼𝘂𝗿𝗰𝗲 𝗣𝗵𝗮𝗿𝗺𝗮𝗰𝗲𝘂𝘁𝗶𝗰𝗮𝗹 𝗜𝗻𝘁𝗲𝗿𝗺𝗲𝗱𝗶𝗮𝘁𝗲𝘀 𝗳𝗿𝗼𝗺 𝗣𝗲𝗽𝗮𝗴𝗼𝗿𝗮?
Pepagora offers a reliable platform for sourcing pharmaceutical intermediates, providing a range of advantages for businesses.
Verified Supplier Network: Pepagora maintains a network of thoroughly vetted suppliers, ensuring that buyers can confidently source high-quality intermediates that meet regulatory requirements.
Streamlined RFQ Process: The platform simplifies the request-for-quotation process, allowing buyers to easily compare offers from various suppliers, saving time and effort.
Trade Assurance and Support: Pepagora provides trade assurance mechanisms that protect buyers throughout the procurement process, ensuring peace of mind with every transaction.
Regional Coverage: With a strong presence in India, the GCC, and Southeast Asia, Pepagora offers extensive regional coverage, facilitating access to a wide array of pharmaceutical intermediates tailored to local market needs.