Similar Products
Description
Pharmalab's Water Systems represent a benchmark in high-purity fluid technology, engineered specifically for the pharmaceutical and biotech industries. These systems utilize advanc...
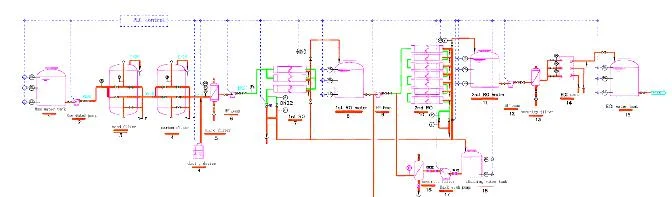
Pharmalab's Water Systems represent a benchmark in high-purity fluid technology, engineered specifically for the pharmaceutical and biotech industries. These systems utilize advanced Thin Film Evaporation distillation to produce Water for Injection (WFI) and Pure Steam, adhering strictly to cGMP, FDA, and ASME guidelines. The design incorporates a robust three-stage pyrogen removal process, specialized centrifugal separation, and an efficient purging mechanism to eliminate impurities, ensuring output that consistently meets the highest pharmacopeial standards. With over three decades of expertise and more than 2500 global installations, these systems are synonymous with reliability, precision, and operational excellence in demanding environments. These water and steam systems are indispensable across the pharmaceutical and life sciences sectors. They are deployed in the manufacturing of injectable drugs, vaccines, biologics, and other sterile products where water and steam quality directly impact product safety. Industries rely on them for critical steps such as sterilizing production equipment, clean-in-place (CIP) systems, and maintaining humidity in aseptic processing areas. Their compliance with international standards makes them suitable for regulated facilities worldwide, ensuring processes from research and development to full-scale commercial production are supported by a trustworthy utility source. The business value of these systems lies in their proven reliability and design integrity, which minimize operational downtime and validation risks. Built for continuous, high-efficiency operation, they offer long-term cost savings through energy-efficient distillation and low maintenance requirements. Their robust construction and adherence to global standards provide peace of mind, reducing regulatory compliance burdens. This reliability translates into consistent product quality, safeguarding your manufacturing output and protecting your brand reputation in markets where purity is paramount. Key Features: - Engineered per cGMP, FDA, USFDA, and ASME standards for regulatory compliance. - Utilizes Thin Film Evaporation distillation technology for superior purity. - Incorporates a unique three-stage pyrogen removal and centrifugal separation system. - Capable of producing both Water for Injection (WFI) and Pure Steam from a single platform. - Features an advanced purging mechanism to eliminate impure water and ensure consistent output quality. Benefits: - Ensures production of high-purity water and steam meeting stringent pharmacopeial requirements. - Enhances process reliability and reduces contamination risk in critical manufacturing steps. - Delivers operational efficiency and long-term cost savings through robust, low-maintenance design. - Supports global regulatory compliance, simplifying validation and audit processes. - Proven track record with over 2500 installations, offering a reliable and trusted solution.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | 106 |
| Customisable | No |
Application
These high-purity water and steam systems are essential for critical pharmaceutical manufacturing processes. They are designed to meet stringent regulatory standards, ensuring the production of Water for Injection (WFI) and Pure Steam that is free from pyrogens and contaminants, which is vital for product safety and efficacy. Reliable and consistent performance makes these systems a cornerstone in facilities where sterility is non-negotiable. They integrate seamlessly into existing production lines, providing a dependable source of pure steam for sterilization and high-grade water for formulation, thereby supporting continuous and compliant manufacturing operations. - Producing Water for Injection (WFI) for parenteral and other sterile pharmaceutical formulations. - Generating Pure Steam for sterilizing tanks, pipelines, and manufacturing equipment in cleanrooms. - Supplying steam for autoclaves and sterilizers used in product and component sterilization processes. - Providing humidification within controlled environments like pharmaceutical cleanrooms and aseptic filling areas. - Cleaning and rinsing critical manufacturing equipment, vessels, and components to prevent contamination.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pharmalab Engineering (India) Ltd, Maharashtra
Industrial Equipment & Machinery•Manufacturer
Factory Details
Factory SizeBelow 1000 sqm
Pharmaceutical Water For Injection And Pure Steam Systems
Pharmalab's Water for Injection and Pure Steam systems are engineered per cGMP, FDA, and ASME standards using Thin Film Evaporation technology for high-purity pharmaceutical applications.
Min. Order Quantity: 1 Pieces
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pharmalab Engineering (India) Ltd
Industrial Equipment & MachineryManufacturer