Similar Products
Description
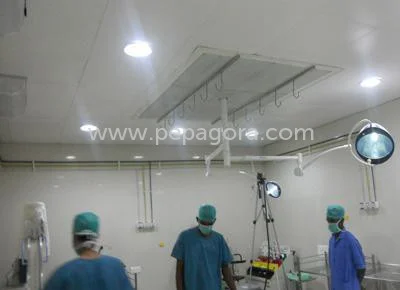
The Lab Pharmaceutical Electronics Vertical Laminar Air Flow System with ISO 5 HEPA filtration represents precision engineering for contamination control in critical environments. ...
The Lab Pharmaceutical Electronics Vertical Laminar Air Flow System with ISO 5 HEPA filtration represents precision engineering for contamination control in critical environments. This advanced system delivers consistently pure air through high-efficiency particulate air filtration that meets ISO 5 classification standards. The vertical airflow design creates a continuous downward curtain of particle-free air that maintains sterile conditions throughout the entire work area. Constructed with durability as a priority, these systems feature robust materials, easy-to-maintain surfaces, and intuitive operational controls that ensure reliable performance while maximizing operational efficiency. The design focuses on practical functionality that supports demanding professional applications where air purity cannot be compromised. These laminar flow systems play vital roles across industries where maintaining sterile conditions directly impacts product quality and safety. In pharmaceutical manufacturing, they protect drug formulation processes and sterile compounding operations from airborne contaminants. Electronics manufacturers depend on these systems for delicate assembly work where microscopic particles could damage sensitive components and affect product performance. Research laboratories utilize them for tissue culture, media preparation, and various sterility testing procedures. Healthcare facilities employ them in pharmacy compounding areas and medical device manufacturing, while food processing operations use them for quality control testing and packaging where maintaining sterile conditions ensures product safety and regulatory compliance. The operational value of these vertical laminar air flow systems extends beyond basic contamination control to deliver measurable business advantages. These units significantly reduce product loss due to contamination, directly improving manufacturing yield and operational profitability. Their energy-efficient design lowers electricity consumption and operational costs while maintaining optimal performance standards. The reliable construction and minimal maintenance requirements ensure continuous operation in critical environments, reducing downtime and associated production delays. The systems help facilities meet stringent regulatory requirements across international markets with comprehensive compliance documentation and validation support, providing peace of mind for quality-conscious operations. Key Features: - HEPA filtration technology achieving ISO 5 classification air purity - Vertical unidirectional airflow pattern for optimal contamination control - Durable construction with easy-to-clean surfaces for maintenance efficiency - Energy-efficient motor and fan system with operational flexibility - Robust design built for continuous operation in demanding environments Benefits: - Maintains sterile conditions for sensitive processes and products - Reduces contamination-related product loss and improves operational yield - Lowers operational costs through energy-efficient performance - Minimizes maintenance requirements and ensures reliable operation - Supports compliance with industry standards and regulatory requirements
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Vertical laminar air flow systems create controlled environments with ISO 5 air purity through advanced HEPA filtration technology. These units maintain unidirectional downward airflow that effectively removes airborne particles, making them essential for processes where even microscopic contamination could compromise product quality or research integrity. The systems provide consistent protection for both sensitive materials and operators while ensuring compliance with stringent industry standards and regulatory requirements across multiple professional sectors. These laminar flow units serve critical functions in pharmaceutical manufacturing, electronics assembly, and scientific research facilities. Their vertical design optimizes floor space utilization while providing maximum protection for delicate procedures. Engineered for seamless integration into existing workflows, these systems feature durable construction that withstands rigorous daily use in demanding environments. The straightforward operation and maintenance requirements make them practical solutions for facilities requiring reliable contamination control without complex operational overhead. - Pharmaceutical sterile compounding and vaccine preparation areas - Electronics manufacturing for circuit board assembly and inspection - Microbiology laboratories for petri dish incubation and testing - Medical device production and packaging operations - Biotechnology research facilities for tissue culture work
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Automech Systems, Karnataka
Industrial Equipment & Machinery•ManufacturerDistributor / Wholesaler
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Lab Pharmaceutical Electronics Vertical Laminar Air Flow System ISO 5 HEPA
Lab Pharmaceutical Electronics Vertical Laminar Air Flow System delivers ISO 5 HEPA filtration for sterile environments, featuring reliable performance and low maintenance design ideal for contamination-sensitive applications.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Automech Systems
Industrial Equipment & MachineryManufacturer