Similar Products
Description
CPM's Sugar Syrup Manufacturing Plant represents the industry standard for pharmaceutical-grade syrup production systems, specifically designed for creating high-purity oral liquid...
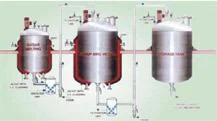
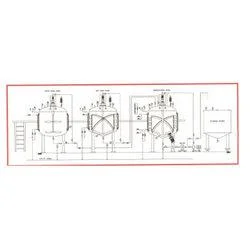
CPM's Sugar Syrup Manufacturing Plant represents the industry standard for pharmaceutical-grade syrup production systems, specifically designed for creating high-purity oral liquids. This advanced plant integrates sophisticated engineering with strict cGMP compliance to deliver exceptional reliability and consistent performance. The system incorporates specialized vessels, precision homogenizers, and zero hold-up filtration technology to maintain product integrity throughout the manufacturing process. With capacity options ranging from 500 to 5000 liters, it accommodates both small-batch development and large-scale commercial production while ensuring uniform quality output with minimal operational variations. This manufacturing plant meets the rigorous demands of pharmaceutical companies, nutraceutical producers, and contract manufacturing organizations globally. It proves particularly valuable for operations producing cough syrups, antacid preparations, vitamin supplements, and pediatric medications where product purity and batch consistency are critical requirements. The system's design specifically addresses the challenges of sugar syrup production, including precise temperature control, homogeneous mixing capabilities, and comprehensive contamination prevention measures. This makes it essential for facilities requiring compliance with FDA, WHO, and other international regulatory standards for oral liquid manufacturing. The operational value of CPM's Sugar Syrup Manufacturing Plant extends well beyond the initial investment through significant reduction in operational costs, minimized product rejection rates, and decreased maintenance requirements. Its proven reliability stems from premium SS316 construction, bottom-entry homogenizers that reduce vibration and maintenance needs, and optional automation features that enhance operational efficiency. The plant's minimal human interface design substantially reduces contamination risks while ensuring product safety and regulatory compliance throughout the equipment's operational lifespan. These features combine to deliver consistent production quality and long-term operational savings. Key Features: - Complete cGMP compliant manufacturing system with SS316 contact parts - Integrated design includes sugar melting, manufacturing, and storage vessels - Bottom-entry stirrer and homogenizer design reduces vibration - Optional CIP/SIP facility and load cell systems available - Semi-automatic or fully automatic PLC operation from 500L to 5000L Benefits: - Ensures regulatory compliance with cGMP and international standards - Minimizes contamination risk through reduced human intervention - Provides consistent product quality across production batches - Reduces operational costs through efficient automation - Offers reliable performance with minimal maintenance requirements
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
CPM's Sugar Syrup Manufacturing Plant is engineered for precise production of high-purity pharmaceutical syrups with automated operation minimizing human intervention. The system ensures consistent quality and reduced contamination risks through advanced homogenization and filtration processes for uniform syrup preparation across various production scales. This plant serves pharmaceutical manufacturers and nutraceutical companies requiring strict hygiene standards and regulatory compliance. It supports semi-automatic to fully automatic PLC operations with optional CIP/SIP systems for sterile processing. The robust SS316 construction guarantees durability while meeting international pharmaceutical manufacturing requirements for oral liquid products. - Production of sugar-based syrups for cough, antacid, and vitamin formulations - Manufacturing of oral rehydration solutions and pediatric medicines - Preparation of base syrups for herbal and Ayurvedic liquid products - Nutraceutical syrup production for health supplements and tonics - Contract manufacturing for branded pharmaceutical oral liquids
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Champion Pharma Machines, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Pharmaceutical Sugar Syrup Manufacturing Plant cGMP Compliant, 500L 5000L
CPM Pharmaceutical Sugar Syrup Manufacturing Plant delivers cGMP compliant oral liquid production with minimal human interface. Available in 500L to 5000L capacities for reliable pharmaceutical syrup manufacturing.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Champion Pharma Machines
Manufacturer