Apparel & Fashion
Loading subcategories...
View More
Description
CPM's Starch Paste Kettle represents precision engineering for pharmaceutical binder preparation, designed to meet strict GMP requirements. This specialized processing unit feature...
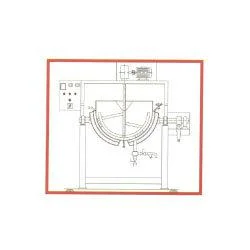
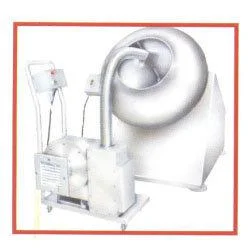
CPM's Starch Paste Kettle represents precision engineering for pharmaceutical binder preparation, designed to meet strict GMP requirements. This specialized processing unit features a hemispherical stainless steel bowl optimized for uniform heat distribution and maximum surface area exposure. The jacketed heating system accommodates multiple energy sources including steam, electrical, or thermic fluid options, all controlled by an adjustable thermostat for precise temperature management. The anchor-type agitator operates through a robust motor and gearbox assembly, ensuring constant stirring that prevents lump formation and achieves perfect paste consistency. With capacities ranging from 10 to 2000 liters and a tilting discharge mechanism, this kettle combines operational efficiency with user-friendly design while maintaining full compliance with pharmaceutical manufacturing standards. This equipment serves critical functions in pharmaceutical manufacturing facilities, particularly in tablet production departments where consistent binder quality directly impacts final product integrity. It is specifically engineered for preparing binder materials that hold active pharmaceutical ingredients together in solid dosage forms. The kettle's pharmaceutical-grade construction makes it suitable for regulated environments where contamination control and material purity are paramount. Beyond core pharmaceutical applications, the unit also finds utility in cosmetic manufacturing for preparing thickeners and in food processing for starch-based formulations requiring precise temperature control. The foundation-free installation allows for flexible deployment in both new facilities and existing production lines undergoing capacity expansion or technology upgrades. The business value of CPM's Starch Paste Kettle lies in its proven reliability and operational efficiency that directly impacts manufacturing outcomes. Manufacturers benefit from reduced batch variation, minimized product loss due to lump formation, and consistent quality in final pharmaceutical products. The equipment's energy-efficient insulation reduces operating costs while the durable stainless steel construction ensures long service life with minimal maintenance requirements. The standardized design ensures compatibility with various production environments while maintaining the precision necessary for pharmaceutical applications. This adaptability makes the kettle suitable for both large-scale manufacturing operations and smaller research applications, providing scalability as production requirements evolve. Key Features: - All contact parts constructed from SS 316 meeting GMP standards for pharmaceutical safety - Hemispherical bowl design with anchor-type impeller for thorough, lump-free mixing - Jacketed heating system compatible with steam, electrical, or thermic fluid energy sources - Motor and gearbox driven agitator with tilting discharge mechanism for efficient operation - Available in capacities from 10 liters to 2000 liters with foundation-free installation Benefits: - Ensures consistent binder quality with uniform heating and mixing for reliable production - Reduces product loss by eliminating lumps and ensuring batch-to-batch consistency - Lowers operating costs through energy-efficient design and minimal maintenance requirements - Enhances workplace safety with tilting mechanism for handling hot materials - Provides flexible installation options suitable for various production environments
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
The CPM Starch Paste Kettle delivers precise binder preparation for pharmaceutical manufacturers requiring consistent, high-quality results. Its hemispherical design and anchor agitator ensure uniform heating and thorough mixing, eliminating lumps and ensuring batch-to-batch consistency. This equipment is essential for producing tablet binders, granulation agents, and coating materials used in solid dosage form manufacturing. Ideal for tablet production environments, this kettle supports steam, electrical, or thermic fluid heating with accurate temperature control. The tilting mechanism enables safe, efficient transfer of hot paste without manual handling. The foundation-free stainless steel framework simplifies installation in existing facilities, making it a versatile solution for expanding production capacity or upgrading older systems. - Preparation of starch-based binders for tablet compression in pharmaceutical manufacturing - Production of granulating agents used in formulating solid oral dosage forms like pills and capsules - Creating coating materials for drug encapsulation and controlled-release formulations - Manufacturing adhesive pastes for applications requiring food-grade or medical-grade materials - Small-scale research and development for developing new pharmaceutical formulations and binder recipes
Trade Details
| Available Stock | In Stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Champion Pharma Machines, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Pharmaceutical Starch Paste Kettle GMP Compliant Binder Preparation
CPM Pharmaceutical Starch Paste Kettle ensures GMP-compliant binder preparation with uniform heating, lump-free mixing, and easy discharge for reliable medicine manufacturing.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Champion Pharma Machines
Manufacturer