Similar Products
Description
The Medical IV Administration Set represents a significant advancement in safe intravenous therapy equipment, featuring PVC and DEHP-free construction that eliminates risks associa...
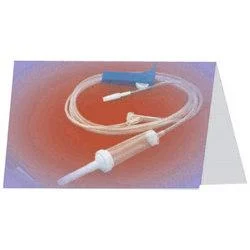
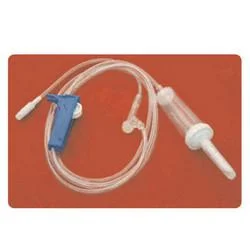
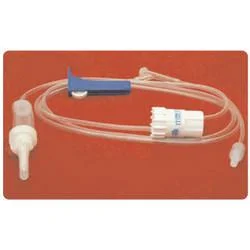
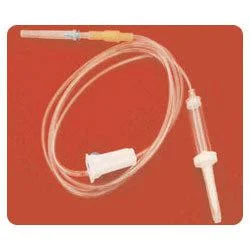
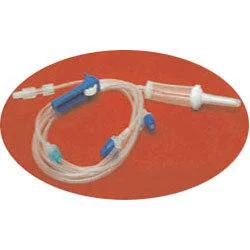
The Medical IV Administration Set represents a significant advancement in safe intravenous therapy equipment, featuring PVC and DEHP-free construction that eliminates risks associated with plasticizer leaching. This sterile medical device incorporates pressure-resistant tubing that maintains structural integrity during high-flow infusions, ensuring consistent fluid delivery without tubing collapse. The clear chamber allows for visual monitoring of fluid flow, while the sharp spike design facilitates easy penetration into IV containers. Manufactured to meet rigorous international standards, this set undergoes gamma or ETO sterilization to guarantee sterility and includes a 15-micron fluid filter that effectively blocks particulate contamination, providing healthcare professionals with a reliable solution for various infusion therapies. Healthcare institutions across multiple medical sectors utilize this administration set for its safety features and compatibility with standard equipment. Hospitals employ it in general wards, surgical units, and intensive care settings for routine hydration and medication administration. Pediatric and neonatal care facilities specifically choose this DEHP-free option to protect vulnerable patients from chemical exposure. Oncology departments rely on it for chemotherapy infusions where material compatibility is critical, while emergency services and ambulatory care centers value its durability and reliability in high-pressure situations. The set's universal connector compatibility makes it suitable for integration into existing medical systems without requiring specialized equipment. The business value of this IV administration set lies in its combination of safety, reliability, and regulatory compliance that directly impacts patient care quality and operational efficiency. Healthcare providers benefit from reduced complication risks associated with plasticizer exposure and particulate contamination, leading to improved patient outcomes and lower treatment costs. The pressure-resistant design ensures uninterrupted therapy during critical procedures, minimizing equipment failure incidents that can compromise patient safety. Compliance with international standards including ISO 8536-4 for spikes and ISO 594 for connectors facilitates seamless adoption across global healthcare markets. These features position this product as a trusted choice for medical distributors seeking to supply high-demand, quality-assured infusion therapy equipment to healthcare facilities worldwide. Key Features: - PVC and DEHP-free tubing and chamber construction for enhanced patient safety - Pressure-resistant tubing design that prevents collapse during high-flow infusions - Sharp spike compliant with ISO 8536-4 standards for secure container penetration - Universal end-connections meeting ISO 594 specifications for equipment compatibility - Integrated 15-micron fluid filter that blocks particulate contamination Benefits: - Eliminates risks of plasticizer leaching for improved patient safety - Maintains consistent fluid delivery during high-pressure infusion therapies - Ensures easy and secure connection to standard IV containers and equipment - Provides universal compatibility with existing medical infusion systems - Prevents particulate contamination during intravenous therapy administration
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
This PVC/DEHP-free IV administration set is engineered for critical care environments where patient safety and precise fluid delivery are essential. Its pressure-resistant tubing maintains consistent performance during high-flow infusions and emergency procedures, while the sterile construction and standardized connectors ensure compatibility with existing medical equipment across various clinical settings. The set's non-toxic, pyrogen-free properties make it suitable for sensitive medical applications where avoiding plasticizer exposure is crucial. Healthcare facilities value its 15-micron fluid filter for preventing particulate contamination during prolonged treatments, providing an additional safety layer for patients receiving intravenous therapies in general medicine, surgery, and specialized care units. - General intravenous fluid and medication administration in hospital wards and intensive care units - High-flow infusion therapy requiring pressure-resistant tubing for consistent delivery rates - Pediatric and neonatal care units where DEHP-free materials are essential for patient safety - Oncology and chemotherapy applications to eliminate risks of chemical leaching - Emergency medical services and trauma care demanding reliable sterile administration sets
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Angiplast Pvt Ltd, Gujarat
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Medical IV Administration Set PVC/DEHP Free Sterile Pressure Resistant
Medical IV Administration Set PVC/DEHP Free Sterile Pressure Resistant delivers safe fluid transfer with ISO-compliant design for hospitals and clinics requiring reliable infusion therapy equipment.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Angiplast Pvt Ltd
Manufacturer