Similar Products
Description
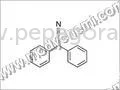
Proginil HCL represents a premium pharmaceutical intermediate engineered for manufacturers and research facilities requiring reliable, high-purity compounds for critical applicatio...
Proginil HCL represents a premium pharmaceutical intermediate engineered for manufacturers and research facilities requiring reliable, high-purity compounds for critical applications. This chemically stable compound delivers consistent performance characteristics essential for precision-driven industries where quality cannot be compromised. Manufactured under stringent quality control protocols, Proginil HCL maintains exceptional structural integrity and purity standards, establishing it as a trusted choice for supply chains demanding technical excellence and batch-to-batch consistency. Its dependable nature ensures manufacturers can integrate it seamlessly into sensitive production processes with confidence in its predictable behavior and performance outcomes. The pharmaceutical manufacturing industry extensively utilizes Proginil HCL in API production, particularly for specialized medications requiring precise chemical architectures and purity profiles. Research institutions and advanced laboratory facilities incorporate this intermediate in developing new therapeutic compounds, conducting experimental studies, and scaling up promising drug candidates. Chemical synthesis operations across global markets rely on Proginil HCL for its consistent reactivity and purity, which directly translates to more predictable manufacturing outcomes and reduced process variability. The compound's reliability makes it suitable for both large-scale production environments and smaller, precision-focused operations where quality consistency is essential for regulatory compliance and successful product development. Selecting Proginil HCL delivers significant business value through reduced production variances and enhanced final product quality. The reliability of this intermediate translates to fewer batch failures, more efficient manufacturing processes, and optimized resource utilization, ultimately protecting profit margins and strengthening customer trust. Its consistent performance characteristics enable manufacturers to maintain tighter control over production timelines, quality assurance protocols, and regulatory documentation. The compound's exceptional stability and extended shelf life ensure distributors and wholesalers can maintain strategic inventory without concerns about degradation, supporting flexible delivery models and minimizing waste throughout the supply chain while ensuring material readiness for production schedules. Key Features: - High-purity pharmaceutical intermediate meeting stringent industry standards and specifications - Consistent molecular structure ensuring reliable and predictable performance in synthesis - Rigorous quality control protocols maintaining exceptional batch uniformity and traceability - Stable chemical composition supporting extended shelf life and storage flexibility - Manufactured under strict quality assurance processes with comprehensive documentation Benefits: - Enhanced manufacturing efficiency through consistent performance and reduced process adjustments - Minimized production variances and batch failures leading to higher yield and output - Improved end-product quality, reliability, and compliance with regulatory requirements - Extended shelf life supporting efficient inventory management and supply chain planning - Trusted performance in critical pharmaceutical processes ensuring production continuity
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Proginil HCL serves as a critical building block in pharmaceutical synthesis, enabling the production of advanced active pharmaceutical ingredients with exceptional purity and stability. Its consistent molecular structure ensures reliable performance in complex manufacturing processes, making it essential for regulated production environments where precision and quality are non-negotiable. This high-purity intermediate is widely utilized across pharmaceutical formulation development, research laboratories, and chemical synthesis operations. Industries value Proginil HCL for its versatility in creating specialized medications and therapeutic compounds, supporting both large-scale commercial production and specialized applications where exacting standards must be consistently met. Real-world applications: - Active pharmaceutical ingredient (API) manufacturing for specialized prescription medications - Research and development of novel therapeutic compounds and drug candidates - Production of high-purity intermediates for regulated pharmaceutical synthesis - Chemical synthesis processes requiring consistent reactivity and precise molecular structure - Formulation development for targeted medications and advanced treatment solutions
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Intermediate Proginil HCL High Purity API Manufacturing
Proginil HCL is a high-purity pharmaceutical intermediate API manufactured for industrial synthesis and medical applications, offering reliable quality and consistent performance for global manufacturing needs.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer