Similar Products
Description
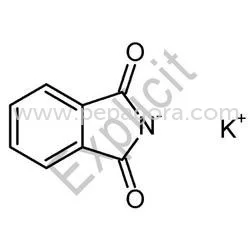
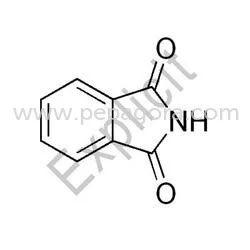
Potassium Phthalimide represents a specialized chemical intermediate renowned for its critical function in organic synthesis, particularly the Gabriel synthesis method for primary ...
Potassium Phthalimide represents a specialized chemical intermediate renowned for its critical function in organic synthesis, particularly the Gabriel synthesis method for primary amine production. This white crystalline powder delivers exceptional purity and consistent reactivity, establishing it as an indispensable component in pharmaceutical manufacturing and fine chemical production. Manufactured under stringent quality control protocols, our Potassium Phthalimide undergoes comprehensive testing for purity, NTU values, and chemical stability to ensure batch-to-batch consistency. The compound is packaged in durable, moisture-resistant materials to maintain integrity during storage and transportation, providing reliable performance for demanding industrial applications. The pharmaceutical industry constitutes the primary application sector for Potassium Phthalimide, where it functions as a key building block for synthesizing active pharmaceutical ingredients in antibiotics, antihypertensive drugs, and central nervous system medications. Chemical manufacturers utilize this intermediate in producing agrochemicals, dyes, and specialty polymers requiring precise amine functionalization. Research institutions and contract manufacturing organizations leverage its consistent quality for developing new compounds and scaling production processes. The compound's reliability makes it particularly valuable for GMP-compliant manufacturing environments where material consistency directly impacts final product quality and regulatory adherence. Businesses select our Potassium Phthalimide for its demonstrated reliability in critical manufacturing processes and consistent quality that minimizes production risks. The product delivers measurable value through enhanced reaction yields, reduced byproduct formation, and improved process efficiency in amine synthesis applications. Our manufacturing process incorporates multiple quality verification points and advanced purification techniques to ensure every batch meets rigorous purity standards. We provide reliable material consistency and technical support to meet specific production requirements, offering flexibility for various industrial applications while maintaining competitive pricing structures. Key Features: - High purity grade suitable for pharmaceutical manufacturing requirements - Consistent reactivity ensuring reliable synthesis outcomes - Comprehensive quality testing including purity and stability parameters - Durable moisture-resistant packaging for optimal product protection - Technical support and consistent batch-to-batch performance Benefits: - Enhanced reaction yields and reduced byproduct formation in synthesis - Reliable performance in critical pharmaceutical manufacturing processes - Consistent quality that minimizes production risks and variations - Optimal storage stability through protective packaging solutions - Improved process efficiency in amine synthesis applications
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Potassium Phthalimide serves as a crucial intermediate in Gabriel synthesis for producing primary amines, a fundamental reaction in organic chemistry. Its high reactivity and stability make it essential for constructing complex molecular structures, particularly in pharmaceutical active ingredients and specialty chemical manufacturing. This compound enables precise and efficient amination processes while minimizing side reactions and improving yields in multi-step syntheses. Industries depend on Potassium Phthalimide for consistent performance in manufacturing antibiotics, antihypertensive medications, and agrochemical intermediates. Its application extends to research laboratories and industrial-scale production where purity and reliability are critical requirements. The compound is particularly valued for processes requiring selective introduction of amino groups, establishing it as a fundamental component in advanced chemical synthesis and fine chemical production. - Synthesis of active pharmaceutical ingredients for antibiotics and cardiovascular medications - Production of agrochemical intermediates including herbicides and pesticides - Manufacturing of dyes and pigments through amine functionalization processes - Research and development in organic chemistry for novel compound creation - Industrial-scale synthesis of specialty chemicals and fine chemical products
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Explicit Chemicals Pvt Ltd, Maharashtra
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Intermediate Potassium Phthalimide For Gabriel Synthesis
Pharmaceutical Intermediate Potassium Phthalimide enables Gabriel Synthesis for primary amine production in bulk drug manufacturing, offering high purity and reliable performance for chemical synthesis applications.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Explicit Chemicals Pvt Ltd
Raw Materials & ChemicalsManufacturer