Similar Products
Description
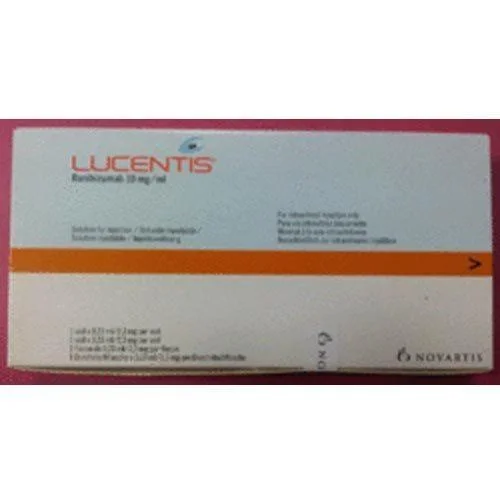
Lucentis ophthalmic injection represents a precision pharmaceutical developed specifically for retinal conditions involving abnormal vascular growth and permeability. The active in...
Lucentis ophthalmic injection represents a precision pharmaceutical developed specifically for retinal conditions involving abnormal vascular growth and permeability. The active ingredient, ranibizumab, is a recombinant humanized monoclonal antibody fragment engineered to selectively bind and neutralize vascular endothelial growth factor-A (VEGF-A). This targeted action addresses the pathological angiogenesis and increased vascular permeability that characterize wet age-related macular degeneration and macular edema. Administered through intravitreal injection by trained ophthalmologists, the treatment follows established protocols beginning with monthly doses, with subsequent frequency adjusted based on individual patient response and disease activity. The formulation ensures optimal therapeutic concentration at the retinal level, making it a fundamental component of contemporary retinal disease management with proven clinical outcomes across multiple studies. This pharmaceutical product serves specialized medical fields including vitreoretinal surgery, medical retina subspecialties, and comprehensive ophthalmology practices. Primary utilization occurs in hospitals with retinal departments, dedicated eye care centers, and specialized retinal clinics where ophthalmologists and retinal specialists diagnose and manage complex retinal pathologies. The medication is particularly valuable in tertiary care institutions that handle advanced retinal cases requiring specialized interventions. Academic medical centers and teaching hospitals also incorporate Lucentis into their therapeutic arsenal for both patient care and educational purposes, training ophthalmology residents and fellows in modern retinal treatment techniques. Additionally, integrated healthcare networks with standardized treatment protocols utilize this product to ensure consistent care quality across multiple facility locations. For medical distributors and procurement specialists, Lucentis offers substantial value through its established clinical profile and consistent market demand. The product maintains exceptional reliability with stringent quality assurance protocols throughout manufacturing, ensuring batch-to-batch consistency in potency and purity. A controlled temperature supply chain preserves product integrity from production through distribution to end-users, minimizing waste and maximizing value for healthcare providers. The documented clinical efficacy creates predictable usage patterns, enabling efficient inventory planning and management for suppliers. Its position as a preferred treatment among retinal specialists ensures sustained demand, making it a stable product category for medical suppliers seeking long-term partnerships with healthcare facilities and maintaining reliable supply to patients who depend on this vision-preserving therapy. Key Features: - Contains ranibizumab, a targeted VEGF-A inhibitor for precise action - Pre-filled syringe format ensures accurate dosing and reduces preparation errors - Manufactured under strict pharmaceutical quality control standards - Temperature-stable formulation simplifies storage and handling requirements - Consistent potency and purity verified through rigorous batch testing Benefits: - Effectively controls abnormal blood vessel growth in retinal conditions - Reduces fluid accumulation in the macula to improve visual outcomes - Pre-filled syringe design enhances administration safety and efficiency - Reliable quality ensures consistent therapeutic performance - Established clinical profile supports confident treatment decisions
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Lucentis injection is a targeted therapy for serious retinal conditions that threaten vision. It contains ranibizumab, which specifically inhibits VEGF-A to reduce abnormal blood vessel growth and fluid leakage in the eye. This mechanism makes it effective for preserving and improving vision in patients with progressive retinal diseases, administered via intravitreal injection by qualified ophthalmologists in clinical settings. This medication addresses two primary indications: wet age-related macular degeneration and macular edema following retinal vein occlusion. For wet AMD, it prevents further vision loss by controlling choroidal neovascularization. For macular edema, it reduces fluid accumulation in the macula to restore visual acuity. Its clinical efficacy is well-documented, making it a standard treatment option in retinal practices and eye care facilities managing these specific conditions. - Monthly intravitreal injections for wet age-related macular degeneration management - Treatment protocol for macular edema following branch retinal vein occlusion - Therapy for macular edema resulting from central retinal vein occlusion - Vision preservation in patients with active choroidal neovascularization - Reduction of retinal fluid accumulation in various macular edema etiologies
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Aark Pharmaceuticals, New Delhi
Trading CompanyDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Ophthalmic Injection Lucentis For Wet AMD And Macular Edema Treatment
Lucentis ophthalmic injection treats wet AMD and macular edema with ranibizumab. This reliable pre-filled syringe offers consistent quality for medical distributors and healthcare providers treating retinal conditions.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Aark Pharmaceuticals
Trading Company