Similar Products
Description
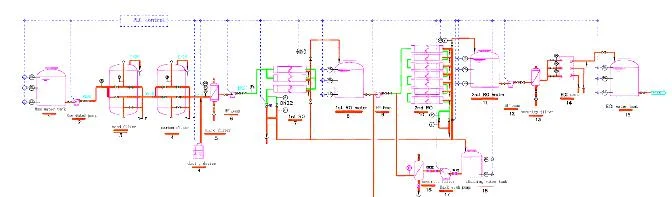
GIECL's High Purity Water System represents a comprehensive water treatment solution engineered specifically for pharmaceutical industries where water quality cannot be compromised...
GIECL's High Purity Water System represents a comprehensive water treatment solution engineered specifically for pharmaceutical industries where water quality cannot be compromised. The system integrates advanced reverse osmosis and electrodeionization technologies to produce water that consistently meets stringent pharmacopeia standards. Utilizing premium US-made components including DOW FilmTec membranes, Grundfoss pumping systems, and Electropure EDI modules, the system ensures reliable performance and consistent output of USP, EP, and JP compliant water. The engineering addresses varying feed water conditions and production capacities while maintaining operational efficiency and compliance with regulatory guidelines throughout the system lifecycle. The pharmaceutical industry depends on high purity water systems for numerous critical applications throughout drug manufacturing processes. These systems are essential for producing Water for Injection used in parenteral formulations, purified water for oral solutions and cleaning validation, and pure steam for sterilization applications. Beyond drug manufacturing, they serve biotechnology companies for cell culture media preparation, API manufacturers for chemical synthesis processes, and medical device companies for rinsing and cleaning applications. The system's robust design ensures continuous operation in GMP-compliant environments, supporting everything from research laboratories to large-scale production facilities with varying water purity requirements and production volumes. Investing in GIECL's High Purity Water System delivers significant business value through enhanced operational reliability, reduced compliance risks, and long-term cost savings. The system's built-in redundancy and automated controls minimize downtime while ensuring consistent water quality that meets evolving regulatory standards. The use of premium components reduces maintenance requirements and extends system lifespan, providing excellent return on investment. The engineering approach means each system is designed for your specific production needs, water source characteristics, and facility layout, ensuring optimal performance and seamless integration with existing manufacturing processes while maintaining regulatory compliance. Key Features: - Advanced RO and EDI technology for consistent ultra-pure water production - Premium US-made components including DOW FilmTec membranes and Grundfoss pumps - Engineered to meet specific pharmaceutical water purity requirements - Automated control systems with continuous monitoring capabilities - Constructed with 316L stainless steel and compliant piping materials Benefits: - Consistent production of pharmacopeia-compliant water quality - Reduced operational risks through reliable US component performance - Lower maintenance requirements and extended system lifespan - Automated operation minimizes manual intervention and human error - Compliance with pharmaceutical industry regulations and standards
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Our high purity water systems deliver critical pharmaceutical-grade water where quality directly impacts product safety and regulatory compliance. These systems consistently produce USP, EP, and JP grade water for injection, purified water, and pure steam essential for manufacturing processes across multiple industries. The robust engineering supports continuous operation in demanding environments while meeting stringent quality standards and ensuring batch consistency. Pharmaceutical manufacturing, biotechnology facilities, API production plants, and parenteral drug manufacturers rely on these advanced water systems. The technology addresses varying feed water conditions and production capacities while maintaining operational efficiency and compliance with current good manufacturing practices. This ensures reliable performance in critical applications where water purity cannot be compromised. - Production of Water for Injection for parenteral and injectable drug formulations - Purified water generation for liquid oral medications and cleaning processes - High purity water supply for biotechnology and cell culture applications - Active Pharmaceutical Ingredient manufacturing requiring ultra-pure water quality - Pure steam production for sterilization and humidification in controlled environments
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Ion Exchange And Chemicals Ltd, Gujarat
Manufacturer
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Pharmaceutical High Purity Water Systems RO EDI US Components
GIECL pharmaceutical high purity water systems feature RO and EDI technology with US components for reliable water treatment in drug manufacturing applications.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Ion Exchange And Chemicals Ltd
Manufacturer