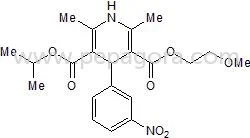
Similar Products
Description
Fluconazole is a potent triazole antifungal active pharmaceutical ingredient used globally in manufacturing medications that combat serious fungal infections. As a trusted supplier...
Fluconazole is a potent triazole antifungal active pharmaceutical ingredient used globally in manufacturing medications that combat serious fungal infections. As a trusted supplier, we provide high-purity Fluconazole in multiple pharmacopeial grades including IP, BP, EP, and USP standards, with additional availability in injectable grade for parenteral formulations. Our products originate from FDA-approved facilities, ensuring regulatory compliance and consistent quality for pharmaceutical manufacturers and distributors requiring reliable bulk API sourcing with complete documentation and certificates of analysis. The pharmaceutical industry extensively utilizes Fluconazole API in antifungal drug manufacturing across multiple therapeutic segments. Hospital pharmacies employ it for compounding medications, while large pharmaceutical companies incorporate it into commercial tablet, capsule, and injection production. Export-oriented manufacturers particularly value our multi-compendial grades which meet stringent international regulatory requirements for markets in North America, Europe, and Asia-Pacific regions where antifungal medications maintain continuous demand. Businesses select our Fluconazole API for its proven reliability, regulatory compliance, and supply chain consistency. We maintain ready stock availability to ensure uninterrupted production schedules, while our FDA-approved status provides the quality assurance that pharmaceutical companies require when sourcing critical APIs for essential medications. Our comprehensive documentation support and batch-to-batch consistency make us the preferred partner for serious buyers seeking dependable antifungal API supplies. Key Features: - Available in IP, BP, EP, USP, and injectable grades for formulation flexibility - Sourced from FDA-approved manufacturing facilities ensuring quality standards - Ready stock availability for immediate shipment and production planning - Comprehensive documentation including certificates of analysis and compliance - Multiple pharmacopeial grade options for international market requirements Benefits: - Ensures regulatory compliance across global pharmaceutical markets - Provides consistent quality and reliable performance in formulations - Supports uninterrupted manufacturing with immediate availability - Reduces compliance risks with complete documentation support - Offers formulation versatility across multiple dosage forms and applications
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Fluconazole API is a broad-spectrum antifungal essential for formulating medications that combat serious fungal infections. It effectively inhibits fungal growth, making it crucial for treating systemic conditions like candidiasis and cryptococcal meningitis. This high-purity compound is integral to producing tablets, injections, and oral suspensions used across healthcare facilities globally. Our Fluconazole meets IP, BP, EP, and USP pharmacopeial standards, ensuring compliance with international regulatory requirements. Pharmaceutical manufacturers and distributors benefit from consistent quality, bulk availability, and complete documentation support for producing various antifungal dosage forms suitable for different patient needs and market regulations. - Manufacturing oral tablets for vaginal yeast infections and oral thrush treatment - Producing injectable solutions for systemic candidiasis in hospitalized patients - Formulating topical creams for skin and nail fungal infection management - Compounding oral suspensions for pediatric and geriatric patients with swallowing difficulties - Supplying bulk API for export markets requiring USP/EP certification compliance
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
A. S. Joshi & Company, Maharashtra
Distributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Bulk Fluconazole API IP BP EP USP Injectable Antifungal
Premium Fluconazole API in IP BP EP USP injectable grades from FDA-approved supplier for antifungal medication manufacturing with reliable quality and bulk availability.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
A. S. Joshi & Company
Distributor / Wholesaler