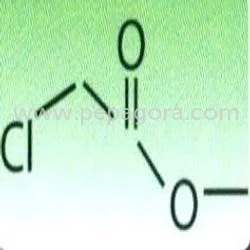
Similar Products
Description
Finasteride bulk active pharmaceutical ingredient represents a high-purity compound essential for manufacturing medications targeting benign prostatic hyperplasia and male pattern ...
Finasteride bulk active pharmaceutical ingredient represents a high-purity compound essential for manufacturing medications targeting benign prostatic hyperplasia and male pattern hair loss. Sourced from an FDA-approved manufacturing facility, this product consistently meets stringent international standards including IP, BP, EP, and USP pharmacopeia grades. With immediate stock availability and decades of manufacturing expertise since 1971, we provide reliable supply chain solutions for pharmaceutical companies, importers, and distributors worldwide. Our commitment to quality assurance and regulatory compliance establishes us as a dependable partner for bulk pharmaceutical ingredients. This API finds extensive application among pharmaceutical manufacturers, compounding pharmacies, and export-focused businesses engaged in generic drug production and specialty formulations. Industries leverage Finasteride in developing oral tablets, capsules, and injectable medications serving urological and dermatological health markets. It addresses global healthcare demands, particularly in regions requiring cost-effective, high-quality active ingredients for therapeutic solutions. Typical users include generic pharmaceutical companies, contract manufacturing organizations, and wholesalers supplying healthcare institutions and retail pharmacies across North America, Europe, and Asian markets. Business partners benefit from our established supply reliability, minimized lead times, and consistent product quality tailored to specific formulation requirements. Our FDA-approved certification and adherence to pharmacopeia standards significantly reduce regulatory compliance risks while ensuring product safety and efficacy. With a legacy of quality manufacturing since 1971, we offer competitive pricing, batch-to-batch consistency, and comprehensive logistical support, creating optimal conditions for sustainable, long-term business relationships. This operational reliability translates into enhanced product trustworthiness and market competitiveness for our commercial partners. Key Features: - Multiple pharmacopeia grade options including IP, BP, EP, USP, and injectable - Sourced from FDA-approved manufacturing facility ensuring compliance - Immediate stock availability for rapid order processing and delivery - Verified high purity and consistency through rigorous quality control - Manufacturing expertise since 1971 guaranteeing product reliability Benefits: - Regulatory compliance simplified through certified manufacturing processes - Supply chain efficiency with ready stock reducing production delays - Consistent quality assurance supporting reliable end-product performance - Global standards compliance facilitating international market access - Established manufacturer track record ensuring partnership stability
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Finasteride API serves as the critical active ingredient in medications addressing androgen-related health conditions. Its mechanism of action involves inhibiting testosterone conversion to DHT, making it essential for formulations targeting urinary flow improvement and hair retention in adult male patients. The compound is manufactured under strict quality protocols to guarantee purity, potency, and batch-to-batch consistency for safe pharmaceutical applications. Pharmaceutical manufacturers and compounding facilities utilize this high-grade API in producing tablets, capsules, and specialized formulations. Suitable for both prescription medications and therapeutic products, it complies with international pharmacopeia standards including IP, BP, EP, and USP. Industries depend on this ingredient to develop effective, scalable treatments that meet regulatory requirements while addressing specific patient healthcare needs across global markets. - Production of prescription medications for benign prostatic hyperplasia treatment - Formulation of hair restoration products including oral and topical solutions - Manufacturing generic pharmaceutical products for affordable healthcare - Supply to compounding pharmacies for customized dosage preparations - Export to international markets for drug development and distribution
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
A. S. Joshi & Company, Maharashtra
Distributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Supplier High Purity Finasteride Bulk API IP BP EP USP
High purity Finasteride bulk API from trusted manufacturer meets IP BP EP USP standards for pharmaceutical production. FDA-approved facility with ready stock ensures reliable supply for global drug manufacturers.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
A. S. Joshi & Company
Distributor / Wholesaler