Similar Products
Description
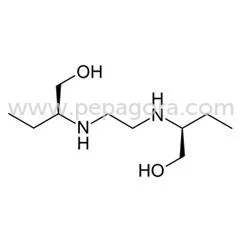
Ethambutol HCL, often referred to as EMB, is a bacteriostatic antimycobacterial medication specifically designed for tuberculosis treatment. As a key component of first-line anti-T...
Ethambutol HCL, often referred to as EMB, is a bacteriostatic antimycobacterial medication specifically designed for tuberculosis treatment. As a key component of first-line anti-TB therapy, it works by inhibiting the synthesis of arabinogalactan in mycobacterial cell walls, effectively halting bacterial replication. With a chemical formula of C10H24N2O2 and a molecular mass of 204.31 g/mol, it maintains stability and efficacy under appropriate storage conditions. Available under trade names such as Myambutol and Servambutol, this pharmaceutical compound adheres to rigorous global quality standards, ensuring reliable performance in healthcare applications. Identified by CAS number 74-55-5 and ATC code J04AK02, it is easily tracked and managed within international pharmaceutical supply chains, providing clarity and consistency for distributors and healthcare providers. This antitubercular agent is vital across various healthcare sectors, including public health initiatives, private hospital networks, and government-led tuberculosis programs. Pharmaceutical distributors supply Ethambutol HCL to treatment centers specializing in respiratory and infectious diseases, where it plays a crucial role in Directly Observed Treatment, Short-course (DOTS) programs, especially in regions with high TB prevalence. International health organizations and medical procurement agencies consistently include this medication in their essential drugs lists, making it a fundamental product for suppliers serving healthcare markets in both developed and developing countries. Its broad application supports efforts to reduce TB incidence and improve patient outcomes worldwide. Businesses that choose our Ethambutol HCL benefit from unwavering quality assurance and dependable supply chain partnerships. We prioritize verified purity levels and batch-to-batch consistency, backed by comprehensive documentation such as Certificates of Analysis, to ensure compliance with international pharmaceutical standards. This focus on reliability and quality differentiation minimizes procurement risks and enhances patient safety, providing distributors and healthcare suppliers with a trusted source for this essential medication. Our commitment to excellence supports seamless integration into treatment protocols, fostering confidence and long-term collaboration in the fight against tuberculosis. Key Features: - Bacteriostatic action targeting tuberculosis-causing mycobacteria - Chemical composition C10H24N2O2 with 204.31 g/mol molecular mass - CAS number 74-55-5 for accurate identification and tracking - ATC classification J04AK02 aligning with international standards - Designed for compatibility with combination therapy approaches Benefits: - Effectively inhibits mycobacterial growth in tuberculosis treatment - Ensures consistent quality and stability for reliable therapeutic outcomes - Facilitates easy identification and integration into global supply chains - Supports standardized treatment protocols to enhance efficacy - Reduces resistance risks when used in multi-drug regimens
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Ethambutol HCL serves as a first-line treatment for pulmonary and extrapulmonary tuberculosis, inhibiting mycobacterial cell wall synthesis to combat actively growing organisms. It is administered in combination with other antitubercular drugs to prevent resistance and ensure effective treatment across diverse patient groups, following established medical guidelines for dosage and duration. Healthcare providers and pharmaceutical distributors depend on Ethambutol HCL for its consistent performance in multi-drug therapy regimens. Suitable for both adult and pediatric cases, it is widely used in hospitals, clinics, and treatment centers due to its proven efficacy and alignment with standard TB treatment protocols, supporting comprehensive care in various healthcare settings. - First-line combination therapy for active tuberculosis in hospital and clinical environments - Management of Mycobacterium avium complex infections in immunocompromised individuals - Pediatric tuberculosis treatment under careful medical supervision - Preventive therapy for latent tuberculosis infections in high-risk groups - Inclusion in multi-drug resistant TB regimens when susceptibility is verified
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Ethambutol HCL Tuberculosis Treatment Bacteriostatic Antimycobacterial
Ethambutol HCL is a bacteriostatic antimycobacterial drug for tuberculosis treatment, ensuring reliable quality for pharmaceutical distributors and healthcare suppliers in global TB control programs.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer