Similar Products
Description
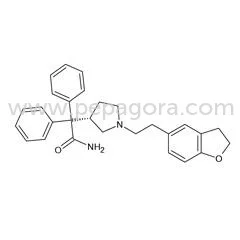
Darifenacin API represents a specialized high-purity active pharmaceutical ingredient engineered for selective muscarinic receptor blockade. This advanced compound specifically tar...
Darifenacin API represents a specialized high-purity active pharmaceutical ingredient engineered for selective muscarinic receptor blockade. This advanced compound specifically targets M3 muscarinic acetylcholine receptors located in the detrusor muscle of the bladder, effectively inhibiting involuntary bladder contractions that cause overactive bladder symptoms. The API demonstrates exceptional bioavailability and consistent pharmacokinetic properties, making it ideal for formulated medications. Its selective action minimizes anticholinergic effects on other organs, providing a superior safety profile compared to non-selective anticholinergics. Pharmaceutical manufacturers worldwide rely on darifenacin for developing targeted therapies that address urinary urgency, frequency, and incontinence with precision and proven clinical efficacy. This API finds extensive application within the urological pharmaceutical manufacturing sector, particularly in medications designed for comprehensive overactive bladder management. Leading pharmaceutical companies incorporate darifenacin into their product lines for geriatric care solutions, specialized health formulations, and chronic condition management drugs. The compound serves as the foundational element for prescription medications distributed through hospital networks, retail pharmacy chains, and specialized healthcare providers. Manufacturing facilities producing extended-release tablets and capsules utilize darifenacin as their primary active component, ensuring consistent therapeutic outcomes and patient satisfaction across global markets while meeting strict regulatory standards including FDA and EMA compliance requirements. Businesses selecting darifenacin API benefit from established reliability and manufacturing consistency that significantly reduces production risks and quality concerns. The compound's well-documented clinical profile and proven efficacy make it an invaluable component for pharmaceutical companies developing competitive products in the urological therapeutics market. Consistent quality assurance processes and supply chain stability ensure uninterrupted production schedules and reliable product availability. The ingredient's selective mechanism provides a distinct competitive advantage in markets where patients and healthcare providers seek targeted therapies with minimized side effects, ultimately enhancing brand reputation and building lasting trust in final pharmaceutical products. Key Features: - Selective M3 muscarinic receptor blockade for targeted therapeutic action - High purity levels consistently meeting international pharmacopeia standards - Reliable bioavailability maintained across all manufacturing batches - Exceptional stability under various formulation and storage conditions - Consistent particle size distribution and optimized solubility profiles Benefits: - Targeted bladder symptom relief with minimal systemic side effects - Compliance with global regulatory standards for pharmaceutical ingredients - Predictable performance in final drug formulations and delivery systems - Extended shelf life and formulation versatility for product development - Manufacturing efficiency through consistent quality and reliable supply
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Darifenacin API serves as the primary active component in advanced pharmaceutical formulations targeting urological disorders. Its selective mechanism specifically blocks M3 muscarinic receptors in bladder muscles, reducing involuntary contractions and providing controlled therapeutic effects for patients experiencing overactive bladder symptoms including urgency and frequency. This high-purity ingredient is essential for manufacturing extended-release tablets and capsules requiring consistent bioavailability and stability. Pharmaceutical manufacturers value darifenacin for its targeted action that minimizes systemic side effects while delivering reliable relief for urinary incontinence and related symptoms across various patient demographics and treatment protocols. - Primary active ingredient in overactive bladder medication formulations - Manufacturing extended-release tablets for sustained therapeutic effect - Production of prescription drugs for urge urinary incontinence - Development of urinary frequency reduction medications - Creating specialized formulations for geriatric care pharmaceuticals
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Darifenacin API High Purity M3 Muscarinic Bladder Treatment
Darifenacin API high purity M3 muscarinic bladder treatment effectively manages overactive bladder symptoms with selective receptor blocking and proven clinical reliability for pharmaceutical manufacturing.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer