Similar Products
Description
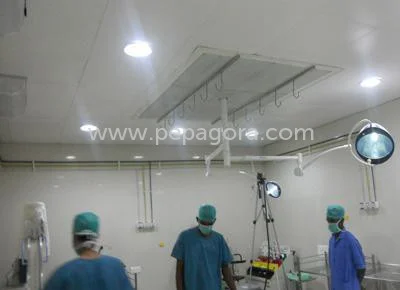
The Class 1000 Clean Room represents advanced contamination control technology designed for industries where air quality and particulate management are critical to operational succ...
The Class 1000 Clean Room represents advanced contamination control technology designed for industries where air quality and particulate management are critical to operational success. This sophisticated clean room system maintains a controlled environment with no more than 1,000 particles (0.5 microns or larger) per cubic foot of air, ensuring exceptional cleanliness standards. The temperature-controlled design provides stable thermal conditions essential for sensitive manufacturing processes, while the room-within-a-room architecture offers enhanced isolation from external contaminants. Every component, from the HEPA filtration systems to the seamless wall surfaces, has been engineered to meet demanding regulatory requirements and industry standards for contamination-sensitive environments. This clean room system serves vital functions across multiple precision industries where contamination control is non-negotiable. Medical device manufacturers rely on it for producing implants, surgical instruments, and diagnostic equipment under sterile conditions. Pharmaceutical companies utilize these facilities for drug formulation, packaging, and sterile filling operations where product purity directly impacts patient safety. The electronics industry depends on these controlled environments for semiconductor production, circuit board assembly, and microcomponent manufacturing where microscopic particles can cause catastrophic product failures. Biotechnology firms employ these clean rooms for cell culture work, genetic research, and vaccine development requiring uncontaminated conditions. The business value of our Class 1000 Clean Room extends beyond regulatory compliance to deliver tangible operational advantages. This contamination control solution represents a strategic investment in quality assurance, risk mitigation, and manufacturing consistency. By implementing this reliable system, businesses significantly reduce product rejection rates, minimize recall risks, and enhance overall production quality. The proven reliability ensures uninterrupted operations while the standardized design provides consistent performance across various applications. This clean room solution delivers long-term value through reduced maintenance requirements, energy-efficient operation, and straightforward integration into existing facilities. Key Features: - Class 1000 certification maintaining ≤1,000 particles (0.5μm) per cubic foot - Precision temperature and humidity control systems for stable environments - Advanced HEPA filtration achieving 99.99% efficiency on 0.3 micron particles - Room-within-a-room design for enhanced isolation and contamination control - Modular construction allowing flexible configuration and easy installation Benefits: - Reduces product contamination risks and improves manufacturing yield - Ensures compliance with industry regulations and quality standards - Provides stable environmental conditions for sensitive processes - Minimizes maintenance requirements and operational downtime - Enhances product quality and reliability through controlled manufacturing environment
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Our Class 1000 Clean Room delivers exceptional contamination control for environments where air quality directly impacts product integrity. It maintains extremely low particulate counts while providing stable temperature and humidity conditions. This makes it essential for manufacturing and research applications where microscopic contaminants can compromise quality, safety, and performance outcomes across multiple high-precision industries. The modular clean room design enables flexible configurations to meet specific operational needs while maintaining consistent performance standards. Featuring advanced air filtration systems, seamless surfaces, and controlled access points, this solution ensures reliable operation in medical devices, pharmaceuticals, biotechnology, and precision electronics manufacturing. The room-within-a-room architecture provides enhanced isolation from external contaminants for superior protection. - Medical device manufacturing and assembly requiring sterile conditions - Pharmaceutical production of drugs and injectables needing contamination-free environments - Microelectronics and semiconductor manufacturing to prevent component damage - Biotechnology research laboratories handling sensitive biological materials - Aerospace component assembly where microscopic particles cause system failures
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Ace Cleanrooms, Maharashtra
ManufacturerDistributor / Wholesaler
Factory Details
Factory Size1000-5000 sqm
Annual Production Capacity0
Main Markets
Domestic
Class 1000 Clean Room For Medical Device And Electronics Manufacturing
Class 1000 Clean Room provides stringent contamination control and precise temperature regulation for medical device manufacturing, pharmaceutical production, and electronics assembly. Advanced HEPA filtration ensures superior air quality.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Ace Cleanrooms
Manufacturer