Similar Products
Description
Industrial Clean-in-Place (CIP) systems represent advanced automated cleaning technology designed for thorough sanitation of processing equipment without manual disassembly. These ...
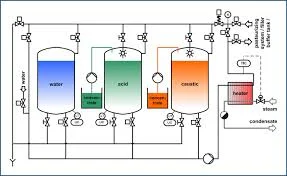
Industrial Clean-in-Place (CIP) systems represent advanced automated cleaning technology designed for thorough sanitation of processing equipment without manual disassembly. These systems utilize precisely controlled sequences of cleaning solutions, applied at optimal temperatures, pressures, and flow rates, to remove product residues, soils, and microbial contaminants from internal surfaces. Engineered for durability and efficiency, CIP technology minimizes water, energy, and chemical consumption while delivering consistent, repeatable cleaning results. The systems are constructed from corrosion-resistant materials suitable for aggressive cleaning environments and are equipped with advanced controls for monitoring critical parameters throughout the cleaning cycle. This combination of robust engineering and intelligent automation ensures reliable performance that meets the stringent hygiene demands of modern industrial operations. CIP systems are indispensable across multiple regulated industries where sanitation directly impacts product safety and quality. In food and beverage manufacturing, they ensure compliance with HACCP and FDA standards by effectively eliminating allergens, pathogens, and biofilms from processing equipment. Pharmaceutical producers rely on CIP technology to maintain sterile manufacturing conditions required by GMP regulations, particularly in active ingredient production and final dosage form manufacturing. Chemical processing facilities utilize these systems to prevent cross-contamination between product batches, ensuring chemical purity and reaction consistency. The dairy industry depends on CIP for daily cleaning of pasteurizers, separators, and storage tanks, while breweries and soft drink manufacturers use them to maintain flavor consistency and prevent microbial spoilage throughout complex piping networks and fermentation vessels. Implementing a CIP system delivers substantial business value through operational efficiency, regulatory compliance, and long-term cost savings. These systems reduce labor requirements by automating cleaning processes that would otherwise require extensive manual intervention, while simultaneously minimizing production downtime between product changeovers or batches. Their reliability stems from robust mechanical construction and sophisticated control systems that monitor and adjust pressure, temperature, chemical concentration, and flow rates in real-time. This automation ensures consistent cleaning outcomes, reduces human error, and provides comprehensive documentation for quality audits. The resulting improvements in equipment uptime, product quality consistency, and regulatory compliance make CIP systems a strategic investment that enhances overall operational resilience and supports business growth in competitive markets. Key Features: - Automated multi-stage cleaning cycles including pre-rinse, detergent wash, acid wash, and final rinse for consistent, repeatable results. - Compatibility with various industrial cleaning agents, sanitizers, and water qualities to suit specific soil removal requirements. - Robust construction using corrosion-resistant materials like stainless steel 304/316 for durability in aggressive chemical environments. - Advanced control systems with programmable logic controllers for precise monitoring and adjustment of temperature, pressure, flow, and concentration. - Customizable design configurations to accommodate specific facility layouts, vessel geometries, and cleaning protocol requirements. Benefits: - Significant reduction in manual labor costs and production downtime through automated cleaning processes between batches. - Enhanced product safety and quality consistency through reliable removal of contaminants, allergens, and microbial hazards. - Improved regulatory compliance with documentation capabilities for HACCP, GMP, FDA, and other quality system requirements. - Optimized consumption of water, energy, and cleaning chemicals through precisely controlled application and recovery systems. - Long-term operational reliability with durable components designed for continuous industrial use and easy maintenance access.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
CIP (Clean-in-Place) systems provide automated, thorough cleaning of industrial process equipment without disassembly. They are engineered to remove residues, contaminants, and microbial growth from tanks, pipelines, and vessels using precisely controlled cycles. This technology is fundamental in industries where hygiene is critical to product safety and quality, significantly reducing manual labor and operational downtime while ensuring consistent sanitation results. These systems are designed to handle various cleaning protocols, including pre-rinse, caustic wash, acid wash, and final rinse stages. They are compatible with a wide range of industrial detergents and sanitizers, making them adaptable to diverse production environments. Built to meet rigorous regulatory standards, CIP systems offer reliable performance that supports continuous operations and helps maintain strict sanitary conditions across different manufacturing scales and layouts. - Food processing facilities for cleaning mixers, conveyors, filling machines, and cooking vessels to prevent allergen cross-contact and pathogen growth. - Dairy plants for sanitizing milk storage tanks, pasteurization units, homogenizers, and cheese vats to maintain product purity and shelf life. - Beverage production lines for maintaining hygiene in brewing kettles, fermentation tanks, bottling machinery, and syrup blending systems. - Pharmaceutical manufacturing suites for ensuring sterile conditions in reactors, blenders, filling lines, and purification equipment to meet GMP requirements. - Chemical processing plants for cleaning reactors, pipelines, and storage vessels between batches to prevent cross-contamination and ensure product integrity.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Anmol Pharma Equipments, Maharashtra
Industrial Equipment & Machinery•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Industrial CIP Systems For Food Beverage Pharmaceutical Cleaning
Industrial CIP Systems for automated cleaning in food, beverage, and pharmaceutical industries. Ensure hygiene, efficiency, and compliance with strict quality standards through reliable, customizable sanitation solutions.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Anmol Pharma Equipments
Industrial Equipment & MachineryManufacturer