Similar Products
Description
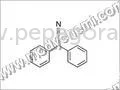
Chlorzoxazone is a high-quality active pharmaceutical ingredient specifically formulated as a muscle relaxant for various medicinal applications. Sourced from our FDA-approved manu...
Chlorzoxazone is a high-quality active pharmaceutical ingredient specifically formulated as a muscle relaxant for various medicinal applications. Sourced from our FDA-approved manufacturing facility with operations dating back to 1971, this API consistently meets international pharmacopeia standards including IP, BP, EP, and USP grades. With ready stock availability, we ensure timely delivery for importers, distributors, and pharmaceutical manufacturers seeking reliable bulk supplies. Our longstanding legacy reflects our commitment to quality assurance and regulatory compliance, establishing us as a trusted partner in the pharmaceutical supply chain for muscle relaxant ingredients. This API finds extensive application across the pharmaceutical industry for manufacturing muscle relaxant medications in various forms including tablets, capsules, and injectable solutions. It effectively addresses musculoskeletal disorders such as muscle spasms, pain, and stiffness in human pharmaceuticals, while also serving veterinary medicine needs for animal musculoskeletal conditions. The ingredient's proven efficacy and safety profile make it indispensable for generic drug manufacturers, hospitals, clinics, and retail pharmacies, providing effective treatment solutions for conditions including back pain, sports injuries, and post-operative recovery scenarios. Businesses benefit from our Chlorzoxazone API through its exceptional reliability, consistent quality assurance, and manufacturing excellence. Our FDA-approved facility and rigorous quality control protocols minimize operational risks while ensuring product consistency batch after batch. The ready stock availability significantly reduces production delays and supports efficient inventory management, enabling manufacturers to maintain smooth production schedules. Our decades-long partnerships since 1971 demonstrate our unwavering commitment to customer satisfaction and trust, providing a competitive advantage in sourcing high-demand pharmaceutical ingredients for global market needs. Key Features: - Complies with multiple pharmacopeia standards including IP, BP, EP, and USP grades. - Manufactured in FDA-approved facility with proven track record since 1971. - Available in ready stock for immediate supply and quick delivery. - Suitable for various formulations including tablets and injectable solutions. - Undergoes rigorous quality testing to ensure purity and therapeutic efficacy. Benefits: - Ensures regulatory compliance with international quality standards. - Reduces supply chain risks through consistent manufacturing quality. - Minimizes production delays with readily available stock. - Supports diverse formulation needs across multiple dosage forms. - Provides reliable therapeutic performance in final medications.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Chlorzoxazone API serves as a centrally acting muscle relaxant in pharmaceutical formulations, effectively treating painful musculoskeletal conditions by blocking nerve impulses in the brain and spinal cord. This mechanism reduces muscle spasms and discomfort, making it essential for producing therapeutic tablets and other dosage forms that provide significant relief to patients suffering from various muscular disorders and injuries. Manufactured under stringent quality control protocols, our Chlorzoxazone complies with IP, BP, EP, and USP standards, ensuring safety and efficacy in final pharmaceutical products. Its consistent quality and broad compatibility make it suitable for diverse formulation needs, supporting seamless production processes and reliable supply chain integration for manufacturers requiring dependable active ingredients for muscle relaxant medications. - Manufacturing muscle relaxant tablets for treating acute back pain and injury-related spasms. - Formulating combination medications that include analgesics for comprehensive pain management. - Producing injectable solutions for hospital use in severe muscle spasm cases. - Supplying bulk APIs to pharmaceutical companies for generic drug production. - Developing veterinary medicines addressing muscle and skeletal disorders in animals.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
A. S. Joshi & Company, Maharashtra
Distributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Chlorzoxazone API Bulk Supplier Muscle Relaxant IP BP EP USP
Chlorzoxazone API muscle relaxant bulk supplier offers IP, BP, EP, USP grades with FDA-approved manufacturing since 1971. Ready stock ensures reliable pharmaceutical sourcing for quality muscle spasm treatments.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
A. S. Joshi & Company
Distributor / Wholesaler