Similar Products
Description
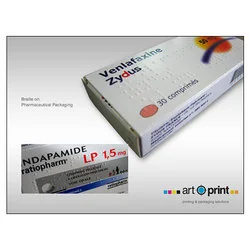
Art O Print delivers specialised braille implementation services for pharmaceutical packaging, addressing critical accessibility requirements in healthcare. Our solution applies br...
Art O Print delivers specialised braille implementation services for pharmaceutical packaging, addressing critical accessibility requirements in healthcare. Our solution applies braille as a separate layer within artwork files, ensuring clear tactile identification while preserving visual design integrity. This service helps pharmaceutical companies meet international regulatory standards and enhances medication safety for visually impaired patients. The process maintains complete separation between braille elements and other design components, enabling precise embossing and thorough quality verification throughout production cycles. This braille implementation service supports pharmaceutical manufacturers, packaging suppliers, and healthcare distributors operating in regulated markets worldwide. The solution proves particularly valuable for companies exporting to European Union countries where braille on pharmaceutical packaging is mandatory. It also serves generic drug manufacturers, clinical research organisations, and hospital pharmacies requiring compliant medication labelling. The service adapts to various packaging formats including cartons, blister packs, medication bottles, and patient information leaflets across both prescription and over-the-counter pharmaceutical products. Art O Print ensures exceptional reliability through rigorous quality control processes and customisation capabilities tailored to specific client requirements. Our approach guarantees that braille implementation never compromises existing packaging designs while maintaining compliance with international accessibility standards. The separate layer methodology facilitates efficient artwork modifications and reduces production timelines. This solution provides pharmaceutical companies with a trusted partner for braille implementation, minimizing compliance risks and enhancing market access across global regions with varying regulatory requirements. Key Features: - Separate braille layer implementation in artwork files - Compliance with international braille standards and regulations - Customisable braille messaging for specific product requirements - Visual verification text outside embossing die area - Colour separation ensuring braille distinctiveness in production Benefits: - Ensures regulatory compliance across global markets - Enhances medication safety for visually impaired users - Maintains brand consistency and packaging integrity - Reduces compliance risks and production timelines - Supports multiple packaging formats and materials
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Pharmaceutical braille labelling ensures medication safety and regulatory compliance across international markets. Art O Print's specialised braille implementation service meets strict accessibility standards while maintaining product integrity and brand consistency. This service is essential for pharmaceutical companies operating in regions with mandatory braille requirements for medicinal products. Our braille solutions integrate seamlessly with existing packaging artwork processes, adding braille as a separate layer without disrupting original design elements. This approach supports efficient visual verification and quality control while accommodating various packaging materials including boxes, leaflets, and blister packs. The service handles multiple languages and global braille standards. - Prescription medication packaging for blind and visually impaired patients - Over-the-counter drug labelling complying with EU accessibility directives - Clinical trial medication packaging with braille identification - Hospital and institutional medication management systems - Export pharmaceutical products to markets with braille regulatory requirements
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Art O Print, Gujarat
Health & Personal Care•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
Domestic
Pharmaceutical Braille Labelling Solutions For Regulatory Compliance
Art O Print provides precise pharmaceutical braille labelling solutions ensuring regulatory compliance and accessibility. Our reliable braille implementation features quality assurance for global pharmaceutical packaging needs.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Art O Print
Health & Personal CareManufacturer