Apparel & Fashion
Loading subcategories...
View More
Description
The Bowie Dick Test Pack represents an essential diagnostic solution specifically engineered to evaluate air removal efficiency in pre-vacuum steam sterilizers. This specialized te...
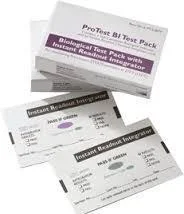
The Bowie Dick Test Pack represents an essential diagnostic solution specifically engineered to evaluate air removal efficiency in pre-vacuum steam sterilizers. This specialized testing system detects residual air pockets that could compromise the sterilization process, ensuring autoclaves operate at optimal performance levels. Designed as a pre-assembled, disposable unit, it simplifies the testing procedure through a distinct color change indicator that transitions from pink to black for clear, unambiguous interpretation. The product maintains compliance with major international standards including AAMI, EN-867-3, and ISO 11140, while incorporating lead-free ink formulations that enhance safety for both users and the environment. This combination of features makes it an ideal choice for facilities that prioritize accuracy, reliability, and ease of use in their sterilization validation protocols. Multiple industries depend on the Bowie Dick Test Pack to uphold their sterility assurance programs and meet regulatory requirements. Healthcare institutions utilize these test packs to validate sterilizers before surgical procedures, ensuring medical instruments are safe for patient use. Pharmaceutical manufacturers integrate them into quality control systems to verify sterile packaging and manufacturing equipment. Research laboratories and biotechnology companies employ these tests to maintain contamination-free environments for sensitive experiments. Additionally, dental practices, veterinary clinics, and medical device manufacturers rely on daily testing to prevent cross-contamination and uphold strict safety protocols across diverse operational settings. The business value delivered by this testing solution centers on risk reduction and operational efficiency. By preventing sterilization failures that could lead to costly product recalls, healthcare-associated infections, or regulatory penalties, organizations achieve significant cost savings and protection of their reputation. The product's reliability stems from consistent performance backed by adherence to globally recognized standards, providing buyers with confidence in their sterilization validation processes. Its user-friendly design minimizes training requirements and reduces potential for human error, while the disposable nature eliminates cleaning and preparation time. These differentiators establish it as a trusted choice for procurement specialists seeking effective, safe, and dependable sterilization testing products that deliver measurable operational benefits. Key Features: - Pre-assembled disposable design ensures convenience and minimizes handling errors - Clear color change indicator from pink to black provides straightforward interpretation - Compliance with AAMI, EN-867-3, and ISO 11140 international standards - Lead-free ink formulation enhances safety for personnel and environmental protection - Specifically engineered for pre-vacuum steam sterilizer air removal validation Benefits: - Reduces sterilization failure risks and associated operational costs - Streamlines compliance with international sterilization standards and regulations - Minimizes training requirements through intuitive design and clear indicators - Enhances patient and product safety through reliable sterilization validation - Saves operational time with ready-to-use, disposable testing format
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
The Bowie Dick Test Pack serves as a critical validation tool for pre-vacuum steam sterilizers, ensuring complete air removal to prevent sterilization failures. Its straightforward color-change indicator provides immediate visual confirmation of sterilizer performance, making it indispensable for maintaining sterile processing standards across medical and laboratory environments where equipment reliability directly impacts safety and compliance outcomes. This testing solution supports daily operational checks in facilities requiring consistent sterilization validation. It helps organizations meet stringent regulatory requirements while minimizing human error through its pre-assembled, disposable design. The pack's compatibility with international standards makes it suitable for global supply chains and diverse operational settings that demand proven sterilization monitoring solutions. - Daily validation of hospital surgical instrument sterilizers before procedures - Routine performance monitoring of dental clinic autoclaves and sterilizers - Quality assurance testing in pharmaceutical manufacturing cleanrooms - Sterilization verification in research laboratories and biotechnology facilities - Compliance monitoring in central sterile supply departments for healthcare accreditation
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Shivaah Packaging, West Bengal
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Bowie Dick Test Pack For Pre Vacuum Steam Sterilizer Air Removal Validation
Bowie Dick Test Pack validates air removal in pre-vacuum steam sterilizers with reliable color-change results for healthcare and laboratory sterilization compliance and safety.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Shivaah Packaging
Manufacturer