Similar Products
Description
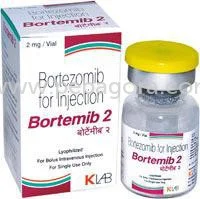
Bortemi-B presents Bortezomib 2mg as a high-purity lyophilized powder within a sterile, single-dose vial, specifically engineered for reconstitution and parenteral administration. ...
Bortemi-B presents Bortezomib 2mg as a high-purity lyophilized powder within a sterile, single-dose vial, specifically engineered for reconstitution and parenteral administration. This formulation prioritizes pharmaceutical precision, offering healthcare providers a trusted solution with consistent bioavailability and exact dosing critical for therapeutic efficacy. Manufactured under stringent quality controls, the product guarantees exceptional purity and stability, extending shelf life and ensuring reliable performance from storage through administration. Its design facilitates safe handling with clear labeling, supporting seamless integration into clinical workflows where accuracy and dependability are non-negotiable for patient safety and treatment success. This product is fundamental within the oncology and hematology sectors, deployed extensively by hospitals, specialized cancer treatment centers, and outpatient clinics. Medical professionals rely on it for managing complex conditions like multiple myeloma and mantle cell lymphoma, where its proven therapeutic profile supports established treatment protocols. Furthermore, pharmaceutical manufacturers, clinical research organizations, and academic laboratories incorporate Bortemi-B into drug development pipelines, clinical trials, and investigative studies, leveraging its reliable composition to advance cancer research and validate new therapeutic approaches. Distributors and wholesalers supply this essential product to a global network of medical facilities, ensuring its availability where it is needed most. The inherent business value of Bortemi-B stems from its uncompromising reliability and precise suitability for high-stakes medical applications. It distinguishes itself through rigorous quality assurance, batch-to-batch consistency, and adherence to international pharmaceutical standards, which collectively minimize operational risk for healthcare providers and distributors. This consistency is paramount for patient safety, predictable treatment outcomes, and maintaining the integrity of clinical research. For supply chain partners, it represents a trusted, in-demand product that enhances inventory reliability and strengthens customer relationships. Its established reputation for therapeutic effectiveness and manufacturing excellence makes it a prudent, low-risk choice for procurement professionals seeking dependable pharmaceutical ingredients. Key Features: - Contains 2mg of the active pharmaceutical ingredient Bortezomib per vial. - Presented as a stable, lyophilized powder for extended shelf life. - Packaged in a sterile, single-use vial for safe reconstitution. - Manufactured to high-purity specifications ensuring consistent composition. - Produced under strict quality control protocols for guaranteed reliability. Benefits: - Enables precise and accurate dosing for critical oncology treatments. - Offers enhanced stability compared to liquid formulations, reducing waste. - Provides a ready-to-use format that integrates smoothly into clinical practice. - Delivers consistent therapeutic performance batch after batch. - Supports patient safety and treatment efficacy through reliable quality.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Bortemi-B is a critical therapeutic agent primarily utilized in hospital oncology and hematology departments for treating multiple myeloma and mantle cell lymphoma. Its lyophilized powder formulation in a sterile vial ensures exceptional stability, precise reconstitution, and accurate dosing, making it indispensable for clinical settings where treatment consistency directly impacts patient outcomes. This reliability supports healthcare professionals in administering effective, standardized care. Beyond direct patient treatment, this product serves pharmaceutical research and development laboratories engaged in advanced cancer therapy studies. Its consistent composition and high purity make it ideal for clinical trials, experimental drug formulations, and investigations into proteasome inhibition mechanisms. The product's dependable quality provides a solid foundation for scientific innovation and the development of next-generation treatment protocols. - Administering precise chemotherapy regimens for multiple myeloma patients in hospital infusion centers. - Treating relapsed or refractory mantle cell lymphoma in specialized oncology clinics. - Serving as a reference standard in pharmaceutical quality control and drug formulation labs. - Supporting clinical research trials for novel combination cancer therapies. - Providing a reliable active pharmaceutical ingredient for compounding pharmacies preparing patient-specific doses.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Khandelwal Laboratories Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerRetailerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Bortezomib 2mg Lyophilized Powder Vial For Oncology Treatment
Bortemi-B Bortezomib 2mg lyophilized powder vial offers high-purity oncology treatment for reliable therapeutic use in medical applications, ensuring consistent quality and precision dosing.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Khandelwal Laboratories Pvt Ltd
Health & Personal CareManufacturer