Apparel & Fashion
Loading subcategories...
View More
Description
Our professional biological indicators for steam sterilization provide the definitive method for validating steam sterilization effectiveness in critical environments. These self-c...
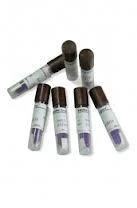
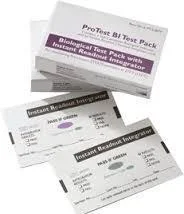
Our professional biological indicators for steam sterilization provide the definitive method for validating steam sterilization effectiveness in critical environments. These self-contained units feature a paper carrier impregnated with Geobacillus stearothermophilus spores, known for their high resistance to steam sterilization. Each indicator is individually packaged in specially designed containers that allow complete steam penetration while maintaining integrity throughout the sterilization process. The integrated culture medium enables straightforward incubation and clear visual reading of results through reliable color change technology, making sterility assurance simple and dependable for technical staff across various applications. These biological indicators serve essential roles across multiple regulated industries where sterility validation is non-negotiable. Healthcare facilities rely on them to validate the sterilization of surgical instruments, implants, and medical devices. Pharmaceutical manufacturers use them to ensure product sterility and manufacturing equipment compliance. Research laboratories depend on them for validating sterile conditions in experimental work, while dental practices employ them to confirm instrument sterility between patient procedures. The food processing industry also utilizes them for equipment sterilization validation, ensuring compliance with health regulations and accreditation standards across all sectors. Organizations choose our biological indicators for their proven reliability and consistent performance in demanding environments. Each batch undergoes rigorous quality control testing to ensure precise resistance values and dependable performance characteristics. The self-contained design eliminates cross-contamination risks and simplifies handling procedures while providing documented evidence of sterilization effectiveness for regulatory compliance and quality audits. The clear color change results minimize interpretation errors, and the robust packaging ensures product integrity during storage and transportation, making them a trustworthy choice for critical sterilization validation requirements. Key Features: - Paper carrier with Geobacillus stearothermophilus spores for reliable resistance - Individually packaged in steam-penetrable, durable containers - Self-contained system with integrated culture medium - Color change technology for easy results interpretation - Manufactured under strict quality control standards Benefits: - Provides definitive validation of sterilization cycle effectiveness - Ensures compliance with international regulatory standards - Reduces risk of healthcare-associated infections - Simplifies sterilization monitoring with clear visual results - Maintains product integrity through robust packaging design
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Professional biological indicators for steam sterilization provide definitive validation of autoclave performance in critical environments. These reliable indicators contain highly resistant bacterial spores that confirm sterilization effectiveness, ensuring patient safety and product sterility across healthcare, pharmaceutical, and research facilities where sterile processing is essential for operational success and regulatory compliance. These biological indicators serve as crucial tools for routine monitoring, load monitoring, and validation of steam sterilization cycles. They are indispensable for meeting international standards like ISO 11138 and AAMI guidelines while helping prevent healthcare-associated infections. Proper implementation ensures the safety of medical devices, pharmaceutical products, and laboratory materials through scientifically proven sterilization verification methods. - Validating steam sterilization cycles in hospital central sterile supply departments - Ensuring sterility of surgical instruments and medical devices before patient procedures - Quality control verification in pharmaceutical manufacturing for sterile drug production - Research laboratories confirming autoclave performance for experimental materials - Dental clinics ensuring complete sterilization of handpieces and surgical instruments
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Shivaah Packaging, West Bengal
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Professional Biological Indicators For Steam Sterilization Validation
Professional biological indicators for steam sterilization validation ensure sterility assurance with reliable paper carriers in steam-penetrable packaging. Trusted by hospitals, labs, and pharmaceutical manufacturers for compliance.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Shivaah Packaging
Manufacturer