Similar Products
Description
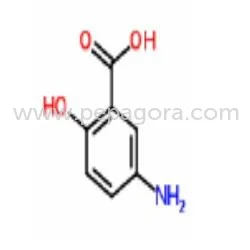
5-Aminosalicylic Acid, identified by CAS 89-57-6 and also known as Mesalazine, is a high-purity pharmaceutical-grade intermediate supplied as an off-white crystalline powder. With ...
5-Aminosalicylic Acid, identified by CAS 89-57-6 and also known as Mesalazine, is a high-purity pharmaceutical-grade intermediate supplied as an off-white crystalline powder. With a molecular formula of C7H7NO3 and molecular weight of 153.14, it features excellent thermal stability with a melting point of 275-280°C (dec.) and boiling point of 403.9°C at 760 mmHg. Its density of 1.491 g/cm3 and flash point of 198.1°C ensure safe handling in industrial pharmaceutical environments. The compound's defined solubility profile and precise chemical structure are fundamental for achieving consistent performance and predictable outcomes in final pharmaceutical formulations, making it a cornerstone material for reliable drug production that meets stringent quality standards. This API is predominantly utilized within pharmaceutical manufacturing for developing and producing medications aimed at chronic inflammatory conditions of the bowel. It serves as the active foundation for various dosage forms including enteric-coated tablets, rectal suppositories, and controlled-release capsules, all designed to deliver therapeutic agents directly to inflammation sites in the colon. Beyond large-scale production, it is vital for research and development laboratories engaged in creating new therapeutic entities, studying disease mechanisms, and improving drug delivery systems for gastrointestinal disorders. Its role is critical in translating research into effective, market-ready treatments that comply with global regulatory standards and pharmacopeial requirements. Selecting this 5-Aminosalicylic Acid provides significant value through assured reliability and quality consistency that aligns with rigorous pharmaceutical standards. Our supply is backed by stringent quality control protocols guaranteeing purity levels essential for API integration, supporting compliance with international pharmacopeial requirements. We prioritize batch-to-batch uniformity to ensure manufacturing processes run smoothly without formulation variances, minimizing production risks and supporting efficient inventory planning for critical drug manufacturing schedules. This reliability in supply and specification ensures continuity for pharmaceutical production lines, making it a trusted source for manufacturers who understand the importance of consistency in their supply chain. Key Features: - Pharmaceutical-grade purity suitable for direct API use in drug manufacturing - Consistent molecular specification with weight of 153.14 for formulation accuracy - High thermal stability evidenced by melting point of 275-280°C (dec.) - Defined physical properties including density and solubility for predictable processing - Supported by comprehensive quality documentation for regulatory compliance Benefits: - Enables reliable production of effective anti-inflammatory medications for bowel diseases - Provides consistent quality that reduces formulation variability and testing time - High stability supports various manufacturing processes and extends shelf-life potential - Well-documented specifications facilitate easier regulatory submissions and audits - Trusted supply ensures continuity for critical pharmaceutical production lines
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
5-Aminosalicylic Acid (Mesalazine) is a critical active pharmaceutical ingredient (API) for producing medications that treat inflammatory bowel diseases. Its high purity and consistent quality make it a trusted component for formulators developing targeted therapies for conditions like ulcerative colitis and Crohn's disease, ensuring therapeutic efficacy and patient safety in final pharmaceutical products. This compound is essential for pharmaceutical manufacturers and research institutions requiring a reliable intermediate for solid and semi-solid dosage forms. It enables the production of formulations designed for specific release profiles and targeted action within the gastrointestinal tract, supporting both established manufacturing processes and innovative drug development pipelines for gastrointestinal disorders. - Core active ingredient in delayed-release tablets for ulcerative colitis treatment - Primary component in rectal suppositories and enemas for localized gastrointestinal therapy - API for manufacturing extended-release oral medications for Crohn's disease management - Key intermediate in research laboratories developing novel anti-inflammatory gastrointestinal drugs - Starting material for synthesizing derivative compounds in advanced pharmaceutical studies
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Yash Rasayan & Chemicals, Gujarat
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Supplier High Purity 5 Aminosalicylic Acid CAS 89 57 6 API
High Purity 5 Aminosalicylic Acid CAS 89-57-6 API is a pharmaceutical-grade Mesalazine intermediate for manufacturing anti-inflammatory bowel disease medications, offering reliable quality and strict compliance.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Yash Rasayan & Chemicals
Manufacturer