Similar Products
Description
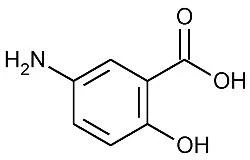
Pharmaceutical Grade 5-Aminosalicylic Acid (5-ASA) represents a high-purity active pharmaceutical ingredient essential for modern gastroenterological treatments. As the active meta...
Pharmaceutical Grade 5-Aminosalicylic Acid (5-ASA) represents a high-purity active pharmaceutical ingredient essential for modern gastroenterological treatments. As the active metabolite of Sulfasalazine, this off-white to pinkish crystalline compound delivers proven anti-inflammatory properties specifically targeting the gastrointestinal tract. Its chemical stability and well-documented pharmacokinetic profile make it a reliable foundation for medications requiring consistent therapeutic outcomes. The compound's sensitivity to moisture and insolubility in water necessitate proper handling protocols, while its acidic nature (approximately pH 4.1 in aqueous solutions) contributes to its targeted action in inflammatory environments. The pharmaceutical manufacturing industry relies heavily on 5-Aminosalicylic Acid for producing medications that treat inflammatory bowel diseases, particularly ulcerative colitis and Crohn's disease. Research laboratories and biotechnology firms utilize this compound for developing novel therapeutic approaches and studying inflammatory pathways. Chemical synthesis operations incorporate 5-ASA as a building block for creating specialized derivatives and fine chemicals. Healthcare providers depend on medications containing this API for managing chronic gastrointestinal conditions, making it a cornerstone of gastroenterological treatment protocols worldwide. Businesses benefit from this Pharmaceutical Grade 5-ASA through its consistent quality, regulatory compliance, and proven therapeutic efficacy. Manufacturers gain reliable supply chain stability with a compound that meets stringent pharmaceutical standards, reducing production risks and ensuring batch-to-batch consistency. The API's established safety profile and extensive clinical documentation provide confidence in product development and regulatory submissions. Its compatibility with various formulation technologies allows for flexible product development across different delivery systems. The compound's targeted mechanism of action translates to effective treatments that support patient outcomes and healthcare provider satisfaction. Key Features: - Pharmaceutical grade purity suitable for medication formulation and regulatory compliance. - Proven anti-inflammatory properties as the active metabolite of Sulfasalazine. - Off-white to pinkish crystalline form with consistent physical characteristics. - Specific storage requirements due to moisture sensitivity and water insolubility. - Acidic aqueous solution profile (approximately pH 4.1) supporting targeted gastrointestinal action. Benefits: - Reliable therapeutic outcomes for inflammatory bowel disease treatments. - Consistent quality meeting stringent pharmaceutical manufacturing standards. - Established safety profile with extensive clinical documentation. - Flexible formulation compatibility across various medication delivery systems. - Targeted action minimizing systemic side effects for improved patient tolerance.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Pharmaceutical Grade 5-Aminosalicylic Acid (5-ASA) serves as the critical active pharmaceutical ingredient in medications designed to manage chronic inflammatory bowel conditions. Its targeted anti-inflammatory action makes it indispensable for formulating treatments that require precise therapeutic effects with high bioavailability and minimal systemic side effects, ensuring patient safety and treatment efficacy. Beyond primary pharmaceutical use, this compound finds application in advanced chemical synthesis and specialized research. Its stable molecular structure and specific reactivity profile make it valuable for developing novel drug derivatives, fine chemical processes, and laboratory reagents where controlled anti-inflammatory properties are essential for scientific and industrial innovation. - Manufacturing oral tablets, capsules, and suppositories for ulcerative colitis and Crohn's disease management. - Formulating topical creams and enemas for localized treatment of gastrointestinal inflammation. - Serving as a key intermediate in synthesizing advanced pharmaceutical compounds and research chemicals. - Supporting clinical research and development of new therapies for autoimmune and inflammatory conditions. - Providing standardized raw material for quality control and analytical testing in pharmaceutical production.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Abhishek Chemicals, Gujarat
Raw Materials & Chemicals•Manufacturer
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Pharmaceutical Grade 5 Aminosalicylic Acid API For Inflammatory Bowel Disease
Pharmaceutical Grade 5 Aminosalicylic Acid API offers high-purity, reliable anti-inflammatory properties for treating inflammatory bowel diseases like ulcerative colitis and Crohn's disease. This active metabolite of Sulfasalazine is essential for gastrointestinal medication formulations.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Abhishek Chemicals
Raw Materials & ChemicalsManufacturer