Similar Products
Description
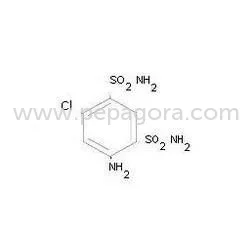
4-Amino-6-Chloro-1,3-Benzene Disulphonamide (CAS 121-30-2) is a high-purity pharmaceutical intermediate manufactured to exacting standards for critical drug synthesis applications....
4-Amino-6-Chloro-1,3-Benzene Disulphonamide (CAS 121-30-2) is a high-purity pharmaceutical intermediate manufactured to exacting standards for critical drug synthesis applications. With a molecular formula of C6H8ClN3O4S2 and a consistent melting range of 254°C to 258°C, this compound delivers reliable, predictable performance in sensitive chemical reactions. Our production process ensures a minimum purity of 99.0%, verified through rigorous quality control protocols including advanced chromatographic analysis. This provides chemical buyers with a consistently dependable raw material that meets international pharmacopeial standards. Proper packaging and comprehensive documentation, including Certificates of Analysis (CoA) and Safety Data Sheets (SDS), support safe handling, traceability, and regulatory compliance throughout the supply chain. This intermediate finds its primary and most significant application in pharmaceutical manufacturing, serving as a crucial precursor in the synthesis of hydrochlorothiazide and other sulfonamide-based diuretics. Manufacturers of cardiovascular medications, bulk drug producers, and API synthesis facilities globally rely on this compound's consistent quality and purity for their core production processes. Research institutions and development laboratories utilize it in projects exploring new therapeutic entities and optimizing synthetic pathways. Furthermore, fine chemical manufacturers incorporate it into specialized formulations where precise molecular structure and high purity are non-negotiable. The compound's inherent reliability and documented quality make it ideally suited for GMP-compliant manufacturing environments and for suppliers serving international markets with strict regulatory requirements. Choosing our 4-Amino-6-Chloro-1,3-Benzene Disulphonamide provides manufacturing operations with a reliably sourced intermediate that ensures exceptional batch-to-batch consistency. This consistency is fundamental to reducing production risks associated with impurity variations, leading to higher synthetic yields, fewer rejected batches, and smoother, more predictable production workflows. The dependable quality directly supports regulatory compliance efforts by providing the necessary documentation and traceability. A stable and reliable supply chain minimizes production downtime and inventory uncertainties. Collectively, these factors enhance end-product quality, improve manufacturing efficiency, and strengthen the market position of companies producing essential medications for both domestic and international healthcare systems. Key Features: - Minimum 99.0% purity, rigorously verified through advanced chromatographic analysis methods. - Precise and consistent melting point range between 254°C to 258°C, ensuring reliable thermal properties. - Compliant with CAS number 121-30-2 for unambiguous international chemical identification. - Supported by comprehensive documentation including Certificates of Analysis (CoA) and Safety Data Sheets (SDS). - Guaranteed batch-to-batch consistency upheld by stringent, multi-stage quality control protocols. Benefits: - Ensures consistent and predictable reaction outcomes in sensitive pharmaceutical synthesis processes. - Significantly reduces production risks and potential batch rejections through guaranteed high purity standards. - Streamlines regulatory compliance and audits with complete, traceable documentation. - Enhances overall manufacturing efficiency and yield by minimizing costly process adjustments and variability. - Provides reliable supply chain support for continuous, uninterrupted production operations.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
4-Amino-6-Chloro-1,3-Benzene Disulphonamide is a critical building block in the synthesis of active pharmaceutical ingredients (APIs), specifically for manufacturing sulfonamide-based diuretic and antihypertensive drugs. Its precise chemical structure and high purity enable controlled, efficient reactions in complex pharmaceutical production, ensuring consistent molecular outcomes that meet stringent pharmacopeial standards for therapeutic efficacy and global regulatory compliance. This specialized intermediate is indispensable for manufacturers producing cardiovascular treatments, generic medications, and fine chemical formulations. It is engineered for use in GMP-compliant production facilities, research laboratories developing new molecular entities, and manufacturing units that require documented quality and batch consistency for reliable supply chains, both domestic and international. - Primary raw material for synthesizing hydrochlorothiazide API in antihypertensive and diuretic drug production. - Key intermediate in manufacturing various sulfonamide-derived cardiovascular medications and related therapeutic compounds. - Essential component for generic pharmaceutical companies producing cost-effective, essential medicines at scale. - Research chemical for pharmaceutical development projects, including molecular structure-activity relationship (SAR) studies. - Fine chemical component in the specialized synthesis of niche therapeutic compounds requiring precise molecular architecture.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Yash Rasayan & Chemicals, Gujarat
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Intermediate 4 Amino 6 Chloro 1,3 Benzene Disulphonamide CAS 121 30 2
High-purity 4-Amino-6-Chloro-1,3-Benzene Disulphonamide CAS 121-30-2 is a 99% min pharmaceutical intermediate essential for synthesizing hydrochlorothiazide API and other sulfonamide-based medications, ensuring reliable supply for manufacturing.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Yash Rasayan & Chemicals
Manufacturer