Apparel & Fashion
Loading subcategories...
View More
Description
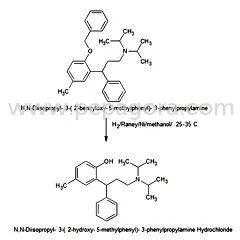
Tolterodine Tartrate is a muscarinic receptor antagonist active pharmaceutical ingredient (API) extensively used in urological therapeutics. With the chemical name (R)-N,N-diisopro...
Tolterodine Tartrate is a muscarinic receptor antagonist active pharmaceutical ingredient (API) extensively used in urological therapeutics. With the chemical name (R)-N,N-diisopropyl-3-(2-hydroxy-5-methylphenyl)-3-phenylpropanamine L-hydrogen tartrate and empirical formula C26H37NO7, it has a molecular weight of 475.6. This white, crystalline powder features a pKa value of 9.87 and water solubility of 12 mg/mL, alongside solubility in methanol, slight solubility in ethanol, and practical insolubility in toluene. Its partition coefficient (Log D) is 1.83 at pH 7.3, indicating optimal bioavailability for pharmaceutical applications. Our product is manufactured under rigorous quality assurance protocols, ensuring high purity and consistency for reliable drug formulation. The pharmaceutical and healthcare industries are the primary users of Tolterodine Tartrate API, particularly manufacturers producing medications for overactive bladder and urinary incontinence. This API is integral to generic drug production, contract manufacturing, and branded pharmaceutical formulations targeting urological health. It is also utilized in clinical research and development for new therapeutic solutions. Industries benefit from its application in tablets, capsules, and controlled-release systems, making it essential for companies focused on geriatric care, urology specialties, and affordable healthcare solutions across global markets. Businesses choosing our Tolterodine Tartrate API gain a reliable, high-quality ingredient that ensures regulatory compliance and end-product efficacy. Our supply chain is optimized for consistency, reducing risks associated with impurities or formulation failures. This reliability translates into faster time-to-market, reduced quality control costs, and enhanced trust with end-users. We differentiate through stringent testing, traceability, and adherence to international standards, providing a competitive edge in regulated markets. Key Features: - High-purity Tolterodine Tartrate API with molecular weight 475.6 - White crystalline powder with optimal solubility characteristics - Partition coefficient (Log D) of 1.83 at pH 7.3 for enhanced bioavailability - Manufactured under strict quality controls ensuring pharmacopeial compliance - Consistent chemical properties including pKa value of 9.87 Benefits: - Reliable performance in urological medication formulations - Ensures regulatory compliance for pharmaceutical manufacturers - Reduces formulation risks through consistent quality and purity - Supports faster time-to-market for drug products - Enhances end-product efficacy and patient outcomes
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Tolterodine Tartrate API serves as the active pharmaceutical ingredient in medications targeting overactive bladder syndrome. It functions by blocking muscarinic receptors in bladder muscle tissue, reducing involuntary contractions and alleviating symptoms including urinary urgency, frequency, and incontinence. This mechanism makes it essential for formulations designed to improve urinary control and enhance patient quality of life across various demographic groups. Manufactured under rigorous quality assurance protocols, our Tolterodine Tartrate meets pharmacopeial standards for purity, stability, and efficacy. Suitable for tablets, capsules, and extended-release formulations, it offers formulation flexibility for pharmaceutical producers. Industries utilizing this API include generic pharmaceutical manufacturers, contract manufacturing organizations, and healthcare suppliers focused on urological and geriatric therapeutic solutions. - Formulation of extended-release tablets for managing overactive bladder symptoms - Production of generic antimuscarinic medications for urinary incontinence treatment - Development of combination therapies for complex urological disorders - Manufacturing prescription medications for geriatric patients with bladder control issues - Creation of specialized formulations for pediatric urinary dysfunction cases
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Grade Tolterodine Tartrate API For Overactive Bladder
Pharmaceutical Grade Tolterodine Tartrate API is a high-purity muscarinic receptor antagonist for urological medications. Manufactured under strict quality controls, it ensures reliable performance in overactive bladder treatment formulations with consistent purity and compliance.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer