Apparel & Fashion
Loading subcategories...
View More
Description
Telmisartan represents a critical angiotensin II receptor blocker active pharmaceutical ingredient specifically developed for effective hypertension management. As a selective anta...
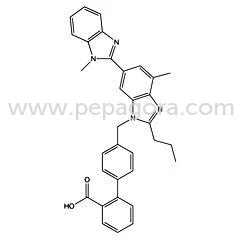
Telmisartan represents a critical angiotensin II receptor blocker active pharmaceutical ingredient specifically developed for effective hypertension management. As a selective antagonist that blocks the binding of angiotensin II to AT1 receptors, it reliably reduces blood pressure by preventing vasoconstriction and aldosterone secretion. Our pharmaceutical-grade Telmisartan meets stringent quality standards with CAS number 144701-48-4 and molecular formula C33H30N4O2, ensuring consistent molecular mass at 514.617 g/mol. This high-purity API enables manufacturers to develop dependable antihypertensive medications that provide sustained 24-hour blood pressure control with single daily dosing, offering superior therapeutic outcomes compared to earlier generation compounds. Pharmaceutical manufacturers across global markets utilize Telmisartan API in producing both branded and generic antihypertensive medications under various established trade names. The cardiovascular pharmaceutical sector particularly values this ingredient for developing advanced combination therapies that address complex hypertension cases requiring multifaceted treatment approaches. Healthcare institutions and medical professionals depend on medications containing Telmisartan for patients needing reliable blood pressure management with minimal side effects and excellent tolerability profiles. The API's compatibility with other cardiovascular agents makes it ideal for fixed-dose combination products that significantly improve patient compliance through simplified treatment regimens and reduced pill burden. Our Telmisartan API delivers exceptional business value through consistent quality, reliable supply chain management, and comprehensive compliance with international pharmacopeial standards. Pharmaceutical partners benefit from our rigorous quality assurance processes that ensure batch-to-batch consistency and purity levels exceeding 99.5%, providing peace of mind for formulation development. The product's excellent stability profile and compatibility with various excipients reduce formulation challenges, accelerating time-to-market for finished pharmaceutical products. With complete documentation including regulatory compliance support, manufacturers receive comprehensive assistance for market approvals across multiple regions, ensuring smooth product launches and sustained market presence. Key Features: - High-purity Telmisartan API with minimum 99.5% purity assurance - CAS number 144701-48-4 compliance with complete traceability documentation - Molecular mass consistency at 514.617 g/mol with precise C33H30N4O2 formulation - Strict adherence to pharmacopeial standards including USP and EP specifications - Excellent stability profile with controlled storage and handling requirements Benefits: - Consistent therapeutic performance through high-purity active ingredient - Complete regulatory compliance support for global market approvals - Reliable batch-to-batch consistency for predictable formulation outcomes - Reduced development time through excellent excipient compatibility - Enhanced product stability ensuring longer shelf life and efficacy
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Telmisartan API serves as the critical active ingredient in antihypertensive medications, specifically formulated to block angiotensin II receptors. This mechanism effectively lowers blood pressure by preventing vasoconstriction, making it essential for cardiovascular disease management. Pharmaceutical manufacturers rely on our high-purity Telmisartan for developing tablets and combination therapies that meet stringent global regulatory standards and ensure patient safety. Our Telmisartan API is manufactured under strict quality control protocols ensuring exceptional purity and batch-to-batch consistency for pharmaceutical formulations. It is suitable for various dosage forms including oral tablets and fixed-dose combinations. The product consistently meets pharmacopeial standards and is ideal for manufacturers producing hypertension medications for diverse markets, providing reliable therapeutic outcomes for patients worldwide. - Formulation of once-daily hypertension tablets for sustained blood pressure control - Production of combination therapies with diuretics for enhanced treatment efficacy - Manufacturing cardiovascular medications for patients with metabolic syndrome - Development of generic antihypertensive drugs for accessible treatment options - Creation of specialized formulations for patients requiring alternative therapies
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Telmisartan API High Purity Hypertension Treatment
High-purity Telmisartan API for hypertension treatment. Premium angiotensin II receptor blocker trusted by pharmaceutical manufacturers for consistent formulation and reliable batch production.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer