Apparel & Fashion
Loading subcategories...
View More
Description
Prochlorperazine Edisylate represents a pharmaceutical-grade sterile injectable solution formulated for precise intramuscular or intravenous administration. Functioning as both an ...
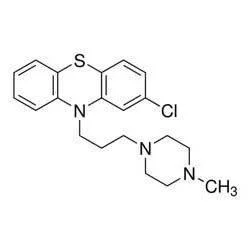
Prochlorperazine Edisylate represents a pharmaceutical-grade sterile injectable solution formulated for precise intramuscular or intravenous administration. Functioning as both an antiemetic and antipsychotic medication, it contains 5 mg of Prochlorperazine per mL as edisylate, carefully compounded with monobasic sodium phosphate monohydrate, sodium tartrate dihydrate, saccharin sodium, and benzyl alcohol in water for injection. The solution operates through targeted depressant action on the chemoreceptor trigger zone, delivering effective control against nausea and vomiting while simultaneously addressing psychotic symptoms. Maintained at a pH between 4.2-6.2, this formulation guarantees stability and clinical compatibility, meeting rigorous quality standards for sterile injectable medications and ensuring reliable therapeutic performance. This medication finds extensive application across multiple healthcare sectors, including hospital systems, oncology treatment centers, psychiatric facilities, and surgical units. In oncology care, it serves as essential supportive medication for managing chemotherapy-induced nausea and vomiting. Psychiatric departments utilize it for rapid intervention in acute psychotic episodes and severe agitation. Surgical wards depend on its antiemetic properties for postoperative recovery management, while emergency departments employ it for acute migraine treatment and other nausea-related emergencies. Its versatility makes it indispensable in medical settings requiring fast-acting injectable formulations for either nausea control or psychiatric stabilization, providing healthcare professionals with a trusted therapeutic option. The value of Prochlorperazine Edisylate lies in its proven reliability, consistent quality standards, and established therapeutic efficacy. Pharmaceutical distributors and healthcare providers can trust its compliance with international quality standards, ensuring patient safety and predictable therapeutic outcomes. The product's demonstrated effectiveness in controlling both emetic and psychotic symptoms makes it a valuable addition to medical inventories. Its sterile formulation and precise chemical composition as 2-Chloro-10-[3-(4-methyl-1-piperazinyl)propyl] phenothiazine 1,2-ethanedisulfonate guarantee batch-to-batch consistency, minimizing operational risks for healthcare facilities and building long-term confidence among medical professionals who depend on consistent medication performance. Key Features: - Sterile injectable solution for intramuscular or intravenous administration - Contains 5 mg Prochlorperazine per mL as edisylate salt - Pharmaceutical-grade excipients ensure formulation stability - pH maintained between 4.2-6.2 for optimal compatibility - Chemical composition: 2-Chloro-10-[3-(4-methyl-1-piperazinyl)propyl] phenothiazine 1,2-ethanedisulfonate Benefits: - Rapid therapeutic action for immediate patient response - Dual functionality addressing both nausea and psychotic symptoms - Consistent batch-to-batch quality and performance - Compatibility with standard medical protocols and procedures - Trusted reliability in diverse clinical environments
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Prochlorperazine Edisylate injectable solution is widely used in clinical environments to control severe nausea and vomiting, particularly in postoperative recovery, chemotherapy-induced nausea, and acute migraine episodes. Its antipsychotic properties make it effective for managing agitation and psychotic symptoms in emergency psychiatric situations where rapid intervention is necessary when oral administration isn't feasible, ensuring quick absorption and predictable therapeutic outcomes. Healthcare facilities including hospitals, oncology centers, and emergency medical services depend on this sterile solution for its consistent performance and safety profile. It serves critical roles in oncology units for chemotherapy support, psychiatric departments for acute symptom management, and surgical wards for postoperative care. The formulation meets strict pharmacopeial standards, ensuring compatibility with diverse medical protocols and patient requirements across various healthcare environments. - Managing chemotherapy-induced nausea and vomiting in cancer treatment centers - Controlling postoperative nausea in surgical recovery and anesthesia care - Treating acute migraine episodes in emergency medical departments - Addressing acute psychotic agitation in psychiatric emergency situations - Providing antiemetic therapy in palliative care and hospice settings
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mehta Pharmaceutical Industries, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Injectable Prochlorperazine Edisylate Anti Nausea Antipsychotic Solution
Injectable Prochlorperazine Edisylate anti-nausea antipsychotic solution offers reliable pharmaceutical-grade quality for managing nausea, vomiting, and psychotic disorders via intramuscular or intravenous administration in clinical settings.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mehta Pharmaceutical Industries
Manufacturer