Apparel & Fashion
Loading subcategories...
View More
Description
Potassium Bitartrate, known chemically as Potassium Hydrogen DL Tartrate, is manufactured as a high-purity white crystalline powder to meet exacting industrial standards. This raw ...
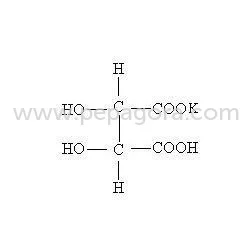
Potassium Bitartrate, known chemically as Potassium Hydrogen DL Tartrate, is manufactured as a high-purity white crystalline powder to meet exacting industrial standards. This raw material is produced with rigorous quality controls to ensure consistent performance in critical applications where precision and reliability are paramount. It is valued for its dual role as an effective acidity regulator and leavening agent, providing stable chemical properties that integrate seamlessly into complex production processes. Manufacturers trust this compound for its predictable behavior and compliance with international safety and purity specifications, making it a foundational ingredient for operations that cannot compromise on material quality. This versatile chemical serves essential functions across multiple industries. In food manufacturing, it is the preferred choice for baking powders, dairy products, and confectionery where precise pH control determines product texture and shelf life. Pharmaceutical producers utilize it as a safe, non-toxic excipient in effervescent tablets and medicinal preparations where consistent dissolution and stability are required. Industrial applications leverage its chemical properties for metal cleaning, electroplating solutions, and as a precursor in tartaric acid synthesis. Its widespread adoption underscores its role as a multifunctional additive that supports operational efficiency, product consistency, and regulatory compliance across diverse production environments. Businesses select this Potassium Bitartrate for its proven reliability and supply chain consistency. The manufacturing process emphasizes batch-to-batch uniformity, reducing production risks and minimizing waste through predictable performance. This reliability translates directly into operational advantages, including consistent end-product quality, reduced rework, and enhanced process efficiency. The compound's stable nature ensures long shelf life and safe handling, while its compliance with international standards simplifies regulatory documentation. By providing a dependable source of this critical raw material, manufacturers gain a strategic partner committed to supporting their production continuity and quality objectives. Key Features: - High-purity white crystalline powder with consistent chemical composition. - Manufactured under strict quality controls for reliable batch-to-batch uniformity. - Stable chemical properties suitable for diverse industrial and food applications. - Compliant with international food safety and pharmaceutical grade standards. - Precise acidity regulation and effective leavening performance characteristics. Benefits: - Ensures consistent product quality and texture in food manufacturing processes. - Provides reliable excipient performance in pharmaceutical tablet formulations. - Reduces production risks through predictable chemical behavior and purity. - Supports regulatory compliance with internationally recognized safety standards. - Enhances operational efficiency by minimizing waste and production variability.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Potassium Bitartrate is a fundamental ingredient in food production, serving as a precise leavening agent in commercial baking powders and a reliable stabilizer in jellies, jams, and processed fruit products. Its consistent quality ensures uniform texture, controlled acidity, and extended shelf life, making it essential for manufacturers who prioritize product consistency and safety in every batch. In pharmaceutical and industrial sectors, this compound provides critical functionality as a safe excipient in effervescent tablets and medicinal preparations. Its chemical properties make it effective for metal cleaning, electroplating solutions, and as a precursor for tartaric acid synthesis, offering versatile performance across technical applications where purity and reliability are non-negotiable. - Essential leavening agent in industrial baking powder for consistent bread and cake production. - Acidity regulator and stabilizer in jams, jellies, and fruit-based beverage manufacturing. - Key excipient in effervescent pharmaceutical tablets and powder formulations. - Effective cleaning and polishing agent for metal surfaces in industrial maintenance. - Primary raw material for synthesizing tartaric acid and derivative chemical compounds.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
National Chemical Industries, Maharashtra
Raw Materials & Chemicals•ManufacturerRetailerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Potassium Bitartrate High Purity Raw Material For Food Pharma
High-purity Potassium Bitartrate raw material for food and pharmaceutical manufacturing. This reliable, consistent-grade powder ensures precise acidity control and leavening performance in industrial applications.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
National Chemical Industries
Raw Materials & ChemicalsManufacturer