Apparel & Fashion
Loading subcategories...
View More
Description
Pharmaceutical Grade Phenylpropanolamine Hydrochloride (PPA HCL) represents a high-purity active pharmaceutical ingredient specifically developed for decongestant applications. Wit...
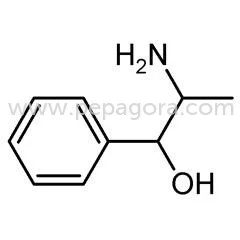
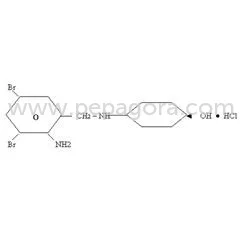
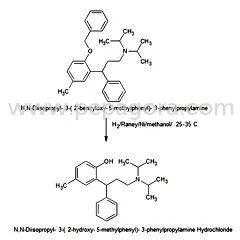
Pharmaceutical Grade Phenylpropanolamine Hydrochloride (PPA HCL) represents a high-purity active pharmaceutical ingredient specifically developed for decongestant applications. With CAS number 14838-15-4 and molecular formula C9H13NO, this compound delivers consistent performance in pharmaceutical formulations targeting respiratory symptoms. Manufactured under stringent quality control protocols, our PPA HCL meets rigorous pharmaceutical standards, ensuring reliability and efficacy for manufacturers developing cold, cough, sinus, and allergy medications. The ingredient's proven track record in pharmaceutical applications makes it a trusted choice for serious manufacturers seeking quality raw materials for their production needs. This versatile API serves multiple industries including human pharmaceuticals, veterinary medicine, and therapeutic product development. In human healthcare, it forms the core component of numerous over-the-counter and prescription decongestants available as tablets, capsules, and syrups. Veterinary applications prominently feature PPA HCL in medications for managing urinary incontinence in dogs, where its pharmacological properties provide effective treatment outcomes. The pharmaceutical industry values this ingredient for its consistent performance and reliability across diverse formulation types, making it essential for companies producing respiratory and allergy relief products. The business value of Pharmaceutical Grade Phenylpropanolamine HCL lies in its consistent quality, reliable performance, and compliance with international pharmaceutical standards. Manufacturers benefit from our rigorous quality assurance processes that ensure batch-to-batch consistency and purity. This reliability translates to reduced production risks and consistent end-product performance, supporting manufacturers in maintaining product quality and regulatory compliance. The ingredient's established efficacy in both human and veterinary applications provides manufacturers with a proven solution for developing effective medications that meet market demands and consumer expectations for relief from congestion and related symptoms. Key Features: - Pharmaceutical grade Phenylpropanolamine HCL with CAS 14838-15-4 - Molecular formula C9H13NO with consistent purity specifications - Manufactured under strict quality control and assurance protocols - Complies with international pharmaceutical standards and specifications - Reliable decongestant properties for respiratory symptom relief Benefits: - High-purity ingredient ensures consistent formulation performance - Proven efficacy in relieving nasal and sinus congestion symptoms - Suitable for diverse pharmaceutical forms including tablets and syrups - Supports regulatory compliance through standardized quality - Trusted reliability for manufacturers requiring consistent API quality
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Phenylpropanolamine HCL serves as a potent decongestant active pharmaceutical ingredient that effectively relieves nasal and sinus congestion by constricting blood vessels. It is widely incorporated into oral medications including tablets, capsules, and syrups designed for respiratory relief. This ingredient maintains consistent performance across various pharmaceutical formulations, making it a trusted choice for manufacturers requiring reliable decongestant action in their products. Beyond human medications, this API finds significant utility in veterinary pharmaceuticals for managing urinary incontinence in animals. It is also utilized in specialized formulations where its pharmacological properties support therapeutic outcomes. Industries relying on this ingredient include pharmaceutical manufacturing, veterinary medicine, and therapeutic product development where quality, purity, and consistent performance are essential requirements for production. - Manufacturing decongestant tablets and capsules for cold and flu symptom relief - Formulating syrups and liquid medications targeting sinus and nasal congestion - Producing combination medications for allergy relief with decongestant properties - Veterinary pharmaceuticals for urinary incontinence management in dogs - Therapeutic formulations requiring reliable vasoconstriction properties
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Grade Phenylpropanolamine HCL API For Decongestants
Pharmaceutical Grade Phenylpropanolamine HCL API is a high-purity decongestant ingredient for cold, cough, sinus, and allergy medications, ensuring reliable performance for manufacturers and distributors.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer