Apparel & Fashion
Loading subcategories...
View More
Description
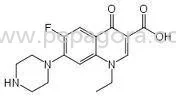
Ondansetron HCL API represents a selective 5-HT3 receptor antagonist recognized as the industry standard for preventing and treating nausea and vomiting in therapeutic applications...
Ondansetron HCL API represents a selective 5-HT3 receptor antagonist recognized as the industry standard for preventing and treating nausea and vomiting in therapeutic applications. Our pharmaceutical-grade active ingredient delivers consistent performance across various drug formulations, manufactured under strict Good Manufacturing Practice guidelines. We provide complete documentation support including certificates of analysis, stability data, and regulatory compliance packages. The product is available in multiple packaging configurations from small R&D batches to commercial production quantities, ensuring operational flexibility for different production scales. Our established supply chain reliability makes us the preferred partner for pharmaceutical companies requiring uninterrupted raw material access for critical medications. The pharmaceutical industry extensively utilizes Ondansetron HCL across multiple therapeutic domains and treatment settings. Oncology departments depend on it for managing chemotherapy-induced nausea and vomiting, while surgical units incorporate it into post-operative care protocols. Gastroenterology specialists integrate it into treatment plans for patients experiencing severe nausea symptoms. Contract manufacturing organizations value our consistent quality for producing generic medications, while research institutions utilize it for developing advanced drug delivery systems. The API serves diverse global markets including regulated regions requiring extensive documentation and emerging markets seeking quality pharmaceutical ingredients. Businesses selecting our Ondansetron HCL gain significant competitive advantages through reliable supply chain partnerships and unwavering product quality. Our manufacturing processes ensure exceptional batch-to-batch consistency, reducing formulation challenges and preventing production delays. The product's compliance with international pharmacopeia standards streamlines regulatory approvals across different markets. Multiple packaging options and comprehensive documentation support simplify import processes for distributors and wholesalers. Long-term partnership benefits include priority allocation during supply constraints and technical assistance for formulation development, creating substantial value beyond standard transactional relationships. Key Features: - Manufactured under strict GMP compliance standards - Comprehensive documentation package with certificates of analysis - Multiple packaging options from kilogram to ton quantities - Consistent particle size and purity specifications - Stability tested with extended shelf life guarantees Benefits: - Ensures regulatory compliance and market approval efficiency - Reduces production delays through batch consistency - Provides formulation flexibility across drug delivery systems - Supports global distribution with complete documentation - Enhances supply chain reliability for critical medications
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Ondansetron HCL API serves as the critical active ingredient in antiemetic medications, specifically formulated to prevent nausea and vomiting associated with chemotherapy, radiation therapy, and surgical procedures. This high-purity pharmaceutical ingredient meets stringent international standards, ensuring compatibility with various drug formulations including tablets, injectables, and oral solutions for global pharmaceutical applications. Pharmaceutical manufacturers and formulation developers rely on our consistent Ondansetron HCL quality for both generic and branded drug production. The product supports diverse therapeutic areas including oncology supportive care, surgical medicine, and gastrointestinal treatments. With flexible batch sizes and comprehensive documentation support, it enables seamless integration into existing manufacturing processes while maintaining regulatory compliance across different markets. - Manufacturing antiemetic tablets for cancer patients undergoing chemotherapy - Formulating injectable solutions for post-operative nausea control in hospitals - Producing oral disintegrating tablets for emergency nausea relief situations - Developing pediatric antiemetic formulations for clinical settings - Creating combination therapies for comprehensive nausea management protocols
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Ondansetron HCL API High Purity Antiemetic Ingredient
Premium Ondansetron HCL API high-purity antiemetic ingredient for pharmaceutical manufacturers. Trusted quality with reliable supply for chemotherapy and post-operative nausea medications. Custom packaging available.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer