Apparel & Fashion
Loading subcategories...
View More
Description
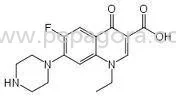
Bavaria Pharma delivers premium Norfloxacin Anti Bacterials API, a high-purity active pharmaceutical ingredient manufactured to exacting pharmaceutical standards. This synthetic fl...
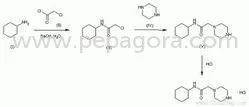
Bavaria Pharma delivers premium Norfloxacin Anti Bacterials API, a high-purity active pharmaceutical ingredient manufactured to exacting pharmaceutical standards. This synthetic fluoroquinolone antibiotic provides reliable broad-spectrum antibacterial activity against numerous pathogenic bacteria, making it an essential component in modern infection control formulations. Our Norfloxacin API undergoes comprehensive quality verification including identity confirmation, purity assessment, and microbiological testing to ensure consistent performance in final pharmaceutical products. The manufacturing process adheres to strict protocols that guarantee pharmaceutical-grade quality batch after batch, providing manufacturers with a dependable active ingredient for their antibacterial medication production needs. This versatile API finds extensive application across pharmaceutical manufacturing sectors, particularly in human medicine for producing antibiotics targeting urinary tract infections, gastrointestinal infections, and sexually transmitted diseases. Veterinary pharmaceutical companies utilize Norfloxacin API in medications for treating bacterial infections in production animals, poultry, and companion pets. Compounding pharmacies incorporate this ingredient into customized medications for patients with specific therapeutic requirements, while research institutions and laboratory suppliers employ it for scientific studies, antimicrobial testing, and formulation development. The product also serves manufacturers developing topical formulations for dermatological applications and ophthalmic preparations for treating eye infections across multiple patient populations. Businesses selecting Bavaria Pharma's Norfloxacin API benefit from established reliability and consistent quality that supports efficient manufacturing operations. Our manufacturing facilities maintain international quality standards, ensuring batch-to-batch consistency and regulatory compliance for diverse global markets. The dependable supply chain minimizes production disruptions for manufacturers, while comprehensive documentation support facilitates regulatory submissions and quality assurance processes. These operational advantages translate to reduced time-to-market for new products, improved end-product therapeutic efficacy, and enhanced satisfaction for healthcare providers and patients relying on effective antibacterial treatments. Key Features: - High purity pharmaceutical grade Norfloxacin API meeting international pharmacopeia standards - Broad-spectrum antibacterial activity effective against Gram-positive and Gram-negative bacteria - Manufacturing under strict quality control protocols with complete documentation support - Consistent particle size distribution and multiple packaging configurations available - Extended shelf life stability with proper storage conditions maintained throughout distribution Benefits: - Ensures reliable therapeutic performance in final pharmaceutical formulations - Provides comprehensive antibacterial coverage for diverse infection types - Facilitates regulatory compliance and quality assurance documentation - Supports efficient manufacturing processes with consistent physical characteristics - Maintains product integrity and potency throughout storage and transportation
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Norfloxacin API serves as a potent broad-spectrum antibacterial agent effective against both Gram-positive and Gram-negative bacteria. Its mechanism inhibits bacterial DNA gyrase, preventing replication and ensuring reliable infection control across multiple therapeutic areas, making it ideal for diverse pharmaceutical formulations requiring consistent antibacterial action and predictable clinical outcomes. This high-purity active pharmaceutical ingredient undergoes stringent quality controls to maintain potency and stability for various dosage forms. Pharmaceutical manufacturers utilize Norfloxacin API for tablets, capsules, ophthalmic solutions, and topical preparations, while veterinary medicine producers incorporate it into animal health products and research laboratories employ it for antimicrobial studies and development projects. - Production of oral tablets and capsules for treating urinary tract infections and gastrointestinal bacterial conditions - Formulation of eye drops and ophthalmic solutions addressing bacterial conjunctivitis and ocular infections - Manufacturing veterinary medications for bacterial infection treatment in livestock and companion animals - Development of topical creams and ointments for skin infections and wound care management - Supply of active ingredient for compounding pharmacies creating customized antibiotic formulations
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Bavaria Pharma Norfloxacin Anti Bacterials API High Purity
Bavaria Pharma Norfloxacin Anti Bacterials API delivers high-purity pharmaceutical-grade active ingredient for manufacturing effective antibacterial medications, trusted globally for quality and reliability.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer