Apparel & Fashion
Loading subcategories...
View More
Description
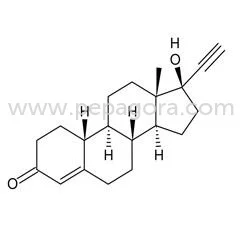
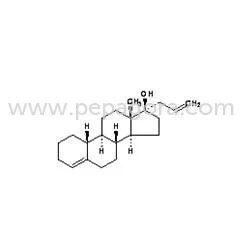
Norethisterone represents a foundational progestin compound in modern pharmaceutical manufacturing, serving as a key active pharmaceutical ingredient in hormonal medications worldw...
Norethisterone represents a foundational progestin compound in modern pharmaceutical manufacturing, serving as a key active pharmaceutical ingredient in hormonal medications worldwide. As a highly active synthetic progestin with exceptional oral bioavailability, it delivers consistent performance across various drug formulations. Our export-grade Norethisterone meets stringent pharmacopeial standards, ensuring purity, potency, and reliability for pharmaceutical manufacturers. The molecule's well-established safety profile and decades of clinical application make it a trusted choice for companies developing contraceptive and therapeutic products. We supply this critical API to licensed pharmaceutical companies and distributors seeking quality raw materials for hormone-based medications with proven efficacy and regulatory acceptance. This versatile progestin finds extensive application across multiple therapeutic areas within the pharmaceutical industry. Major contraceptive manufacturers incorporate Norethisterone into combined oral contraceptive pills, where it works synergistically with estrogen components to provide reliable birth control. Women's health pharmaceutical companies utilize it in progestogen-only pills for patients requiring estrogen-free options due to medical contraindications or personal preferences. Additionally, gynecological medicine producers formulate it into treatments for menstrual disorders including premenstrual syndrome, dysmenorrhea, and menorrhagia. The compound also serves hormone replacement therapy products for menopausal management, demonstrating its broad utility across different medication categories and diverse patient population needs. Pharmaceutical businesses value Norethisterone for its proven reliability and manufacturing flexibility that supports efficient production processes. Our supply chain ensures consistent availability to meet production schedules, while rigorous quality assurance protocols guarantee batch-to-batch consistency that manufacturers can depend on. The compound's chemical stability allows for various formulation approaches, enabling manufacturers to develop both immediate and extended-release products according to market demands. Its established regulatory status in multiple international markets simplifies product registration and compliance processes, reducing administrative burdens. By sourcing through our reliable network, businesses gain access to a critical API with documented efficacy, minimizing development risks and accelerating time-to-market for hormone-based pharmaceutical products. Key Features: - High-purity synthetic progestin meeting international pharmacopeial standards - Excellent oral bioavailability with consistent pharmacokinetic performance - Stable chemical structure suitable for various formulation types and delivery systems - Comprehensive documentation including certificates of analysis and compliance - Reliable supply chain with consistent batch-to-batch quality assurance Benefits: - Proven clinical efficacy supported by decades of therapeutic use - Manufacturing flexibility supporting diverse product development needs - Simplified regulatory compliance due to established international acceptance - Reduced development risks with well-documented safety and performance data - Consistent quality ensuring reliable end-product performance and patient outcomes
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Norethisterone API serves as a critical active pharmaceutical ingredient in hormonal therapeutics, primarily formulated into combined oral contraceptive pills and progestogen-only medications. Its stability and bioavailability make it ideal for tablet and capsule manufacturing, ensuring consistent dosage delivery for patients and reliable performance in various drug delivery systems. Pharmaceutical companies value its proven efficacy and compatibility with diverse formulation requirements, supporting consistent production outcomes. Beyond contraceptive applications, this versatile progestin effectively addresses multiple gynecological and endocrine conditions. It manages premenstrual syndrome, dysmenorrhea, menorrhagia, and irregular menstrual cycles with established clinical success. In menopausal hormone therapy, it combines with estrogen to reduce symptoms while protecting endometrial health. Its ability to postpone menstruation provides valuable flexibility in treatment protocols for special medical and lifestyle needs, making it a multifunctional therapeutic agent. - Active ingredient in combined oral contraceptive pills for effective pregnancy prevention - Core component in progestogen-only pills for specialized birth control formulations - Therapeutic agent for managing heavy menstrual bleeding and menorrhagia conditions - Essential component in hormone replacement therapy for menopausal symptom management - Medical treatment for menstrual cycle regulation and therapeutic period postponement
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Norethisterone API High Purity Progestin Hormone Therapy
High-purity Norethisterone API for pharmaceutical manufacturing. This reliable progestin is essential for oral contraceptives, hormone therapy, and menstrual disorder treatments, ensuring consistent potency and global export compliance.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer