Apparel & Fashion
Loading subcategories...
View More
Description
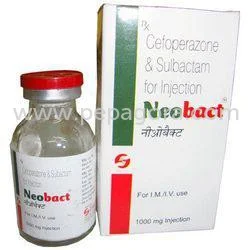
Neobact sterile cefoperazone sodium injection 1000mg represents pharmaceutical excellence in antibiotic therapy, providing cefoperazone sodium IP equivalent to 1000mg anhydrous cef...
Neobact sterile cefoperazone sodium injection 1000mg represents pharmaceutical excellence in antibiotic therapy, providing cefoperazone sodium IP equivalent to 1000mg anhydrous cefoperazone per vial. This third-generation cephalosporin antibiotic delivers broad-spectrum activity against both Gram-positive and Gram-negative bacteria, including many resistant strains that challenge conventional treatments. Manufactured under strict sterile conditions following international pharmacopeia standards, each vial ensures maximum potency and patient safety. The reliable formulation makes it a preferred choice for medical professionals managing serious infections where treatment failure carries significant consequences, offering consistent therapeutic outcomes across diverse clinical scenarios. This injectable antibiotic serves critical needs across multiple healthcare sectors including hospital pharmacies, surgical centers, and critical care facilities. Medical professionals in internal medicine, surgery, and infectious disease specialties regularly utilize this product for treating complicated infections that require immediate and effective intervention. The product supports both government healthcare institutions and private hospital networks, with particular importance in emergency departments and intensive care units where rapid bacterial control is essential. Veterinary medicine applications also benefit from this formulation in large animal practices where injectable antibiotics are required for addressing serious infections in clinical settings. Healthcare providers achieve enhanced therapeutic value through consistent quality assurance and reliable clinical outcomes when using Neobact cefoperazone sodium injection. The product contributes to reduced treatment failure rates and decreased hospital stay durations, supporting better patient outcomes and optimized healthcare resource utilization. The sterile manufacturing process ensures batch-to-batch consistency while eliminating contamination risks during administration. Pharmaceutical distributors appreciate the product's stable shelf life and compliant packaging that maintains integrity throughout transportation and storage. Healthcare institutions benefit from the product's proven efficacy in managing complex infections, supporting their commitment to delivering high-quality patient care. Key Features: - Sterile cefoperazone sodium IP formulation meeting pharmacopeia standards - Equivalent to 1000mg anhydrous cefoperazone per vial for precise dosing - Broad-spectrum antibiotic activity against resistant bacterial strains - Manufactured under strict quality control and sterile conditions - Reliable formulation ensuring consistent therapeutic performance Benefits: - Effective treatment for serious bacterial infections requiring potent therapy - Trusted sterile formulation ensuring patient safety during administration - Broad-spectrum coverage addressing diverse infection types - Consistent quality and reliable clinical outcomes across applications - Stable shelf life and compliant packaging for distribution efficiency
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Neobact sterile cefoperazone sodium injection 1000mg serves as a critical therapeutic option in clinical settings for combating severe bacterial infections. Its broad-spectrum efficacy makes it particularly valuable against resistant pathogens commonly encountered in hospital environments, including those causing complicated intra-abdominal infections and surgical site infections requiring potent parenteral antibiotic therapy. This pharmaceutical injection finds essential application across multiple medical specialties including intensive care, surgery, and internal medicine. Healthcare professionals rely on this sterile formulation for treating respiratory tract infections, urinary tract infections, and skin structure infections where other antibiotics have demonstrated limited effectiveness, ensuring safe administration for immunocompromised patients and those in critical condition. - Hospital-acquired pneumonia treatment in intensive care units - Surgical site infection prevention in complex abdominal procedures - Complicated urinary tract infection management in hospitalized patients - Skin and soft tissue infection treatment for diabetic patients - Febrile neutropenia management in oncology and hematology departments
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Navdeep Bioceuticals, Haryana
Raw Materials & Chemicals•ManufacturerDistributor / Wholesaler
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Cefoperazone Sodium Injection 1000mg Sterile Antibiotic
Neobact sterile cefoperazone sodium injection 1000mg provides reliable broad-spectrum antibiotic treatment for serious bacterial infections, trusted by hospitals and healthcare providers for consistent quality and sterile formulation.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Navdeep Bioceuticals
Raw Materials & ChemicalsManufacturer