Apparel & Fashion
Loading subcategories...
View More
Description
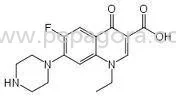
Misoprostol 1% HPMC represents a premium pharmaceutical-grade solution combining high-purity Misoprostol with Hydroxypropyl Methylcellulose to create a stable, bioavailable formula...
Misoprostol 1% HPMC represents a premium pharmaceutical-grade solution combining high-purity Misoprostol with Hydroxypropyl Methylcellulose to create a stable, bioavailable formulation. This specialized solution meets rigorous pharmaceutical standards, ensuring consistent quality and performance for medical and research applications. The HPMC matrix enhances stability and controlled release properties, making it ideal for formulations requiring precise dosage accuracy and long-term reliability. Manufactured under strict quality control protocols, this solution delivers the purity and consistency that pharmaceutical professionals depend on for critical applications where ingredient quality directly impacts end-product effectiveness and safety. This pharmaceutical solution serves multiple industries including pharmaceutical manufacturing, healthcare institutions, research organizations, and veterinary medicine. In pharmaceutical production, it forms the core active component in oral medications, particularly tablets and capsules where dosage precision and stability are crucial. Healthcare facilities utilize it in clinical settings for specialized treatments, while research laboratories rely on its consistent quality for drug development studies and efficacy testing. Veterinary pharmaceutical companies incorporate it into animal health products, benefiting from its reliable performance across different biological systems. The solution's versatility and compliance with industry standards make it valuable across these diverse sectors. The business value of Misoprostol 1% HPMC lies in its unwavering reliability, consistent quality, and formulation integrity that reduces operational risks for purchasing organizations. Its differentiation stems from strict adherence to manufacturing specifications and proven performance in demanding applications, ensuring suitability for high-stakes pharmaceutical production. This reliability fosters trust among supply chain partners, supporting long-term business relationships and minimizing production disruptions. By providing a dependable pharmaceutical ingredient that meets regulatory requirements and performance expectations, it helps businesses maintain competitive positioning, streamline manufacturing processes, and deliver successful end-products to market. Key Features: - Pharmaceutical-grade 1% Misoprostol formulation with HPMC for enhanced stability - Strict manufacturing standards ensuring consistent quality and performance - High-purity composition suitable for sensitive medical applications - Proven bioavailability and efficacy across various delivery systems - Stable formulation maintaining integrity under standard storage conditions Benefits: - Reliable ingredient quality reducing production risks and variability - Consistent performance supporting predictable manufacturing outcomes - Enhanced stability extending product shelf life and usability - Versatile application across pharmaceutical and research settings - Trusted formulation meeting rigorous industry standards and requirements
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Misoprostol 1% HPMC is extensively utilized in pharmaceutical manufacturing processes where precise dosage control and formulation stability are paramount. This pharmaceutical-grade solution maintains consistent performance in tablet and capsule production, meeting stringent quality requirements for therapeutic medications. Its reliable composition ensures uniform distribution in final products, making it suitable for large-scale production environments that demand exacting standards and repeatable results across manufacturing batches. Research institutions and healthcare facilities benefit from this solution's purity and efficacy in clinical studies and treatment protocols. Laboratories utilize it for drug development projects requiring stable active ingredients, while medical centers incorporate it into patient care regimens where dependable pharmaceutical components are essential. The solution's compatibility with various delivery systems and research methodologies makes it valuable across multiple scientific and medical disciplines requiring trusted ingredients. - Pharmaceutical tablet and capsule manufacturing requiring precise active ingredient distribution - Clinical research studies investigating drug efficacy and therapeutic applications - Hospital pharmacy departments for compounding specialized medication formulations - Veterinary pharmaceutical production for animal healthcare products - Academic research institutions conducting pharmacological studies and drug development
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Misoprostol 1% HPMC Pharmaceutical Grade Solution
Misoprostol 1% HPMC pharmaceutical grade solution offers high-purity formulation for medical manufacturing and research applications, ensuring reliable quality and consistent performance.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer