Apparel & Fashion
Loading subcategories...
View More
Description
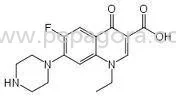
Metoclopramide HCL is a high-purity active pharmaceutical ingredient specifically developed for antiemetic and gastroprokinetic pharmaceutical formulations. This API functions thro...
Metoclopramide HCL is a high-purity active pharmaceutical ingredient specifically developed for antiemetic and gastroprokinetic pharmaceutical formulations. This API functions through dual mechanisms as a dopamine antagonist and serotonin receptor agonist, effectively accelerating gastric emptying while providing reliable control over nausea and vomiting symptoms. Manufactured in GMP-compliant facilities, it consistently meets stringent pharmacopeial standards including USP, EP, and IP specifications. The product demonstrates exceptional stability and purity, making it suitable for various pharmaceutical dosage forms from tablets to injectables. Its consistent quality ensures reliable performance in final drug products, supporting pharmaceutical manufacturers in creating effective medications for patients suffering from gastrointestinal disturbances and treatment-related nausea. This pharmaceutical ingredient finds extensive application across multiple healthcare sectors, particularly in manufacturing medications for gastrointestinal disorders, oncology support care, and surgical recovery protocols. Generic drug manufacturers incorporate Metoclopramide HCL into their product lines for antiemetic tablets and injectables, while hospital pharmacies utilize it for compounding specific patient formulations. The API serves pharmaceutical companies targeting markets in North America, Europe, and Asia, where regulatory compliance and consistent quality are paramount. Its versatility extends to various therapeutic areas including chemotherapy adjunct therapy, postoperative care, and chronic condition management, making it a fundamental component in modern pharmaceutical manufacturing and patient care protocols. Businesses select Metoclopramide HCL for its proven reliability, regulatory compliance, and consistent supply chain performance. The API's batch-to-batch uniformity reduces formulation risks and supports smooth regulatory submissions across international markets. Its manufacturing under strict quality controls ensures every shipment meets specified purity and safety standards, minimizing production delays and quality concerns. The comprehensive documentation package including Certificates of Analysis and stability data provides manufacturers with complete traceability and confidence in their final products. This reliability translates into enhanced end-product efficacy and patient safety, establishing trust between suppliers and pharmaceutical companies while supporting successful market applications and therapeutic outcomes. Key Features: - High purity grade meeting USP, EP, and IP pharmacopeial standards - GMP-compliant manufacturing with rigorous quality assurance protocols - Consistent batch-to-batch performance for formulation reliability - Suitable for tablets, injectables, and oral solution formulations - Comprehensive documentation including COA and stability data Benefits: - Ensures reliable nausea and vomiting control in final medications - Supports regulatory compliance for international market approvals - Reduces formulation risks through consistent quality and performance - Enhances manufacturing efficiency with reliable supply chain - Provides complete traceability and quality assurance documentation
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Metoclopramide HCL serves as a critical active pharmaceutical ingredient in antiemetic and gastroprokinetic drug formulations. It accelerates gastric emptying and effectively controls nausea and vomiting through dopamine antagonist and serotonin agonist activity. Pharmaceutical manufacturers rely on its high purity and consistent performance for developing tablets, injectables, and oral solutions that meet strict pharmacopeial standards and regulatory requirements across global markets. This API supports pharmaceutical companies, compounding pharmacies, and bulk distributors in producing medications for gastrointestinal disorders, chemotherapy support, and postoperative care. Its reliable efficacy makes it suitable for generic drug production, hospital formulations, and export-oriented manufacturing. Industries value its adaptability to various dosage forms and compliance with international quality protocols, ensuring safe and effective patient treatments across diverse therapeutic applications. - Manufacturing antiemetic tablets for chemotherapy-induced nausea and vomiting management - Producing injectable solutions for postoperative and emergency nausea control - Formulating gastroprokinetic medications for diabetic gastroparesis treatment - Compounding oral suspensions for pediatric and geriatric patient populations - Supplying bulk APIs for generic antiemetic drug production in regulated markets
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical API Metoclopramide HCL High Purity Antiemetic Bulk
Metoclopramide HCL high-purity antiemetic API for pharmaceutical manufacturing ensures reliable nausea control with GMP compliance and batch-to-batch consistency for tablets and injectables.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer