Apparel & Fashion
Loading subcategories...
View More
Description
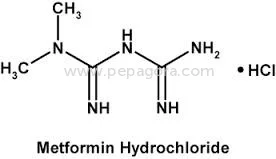
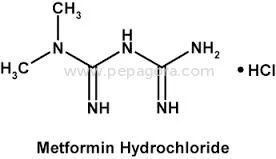
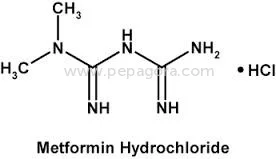
Metformin Hydrochloride represents pharmaceutical-grade excellence with a guaranteed assay of 98.5-101.0% and impurity levels maintained at ≤0.1%, ensuring superior quality for ant...
Metformin Hydrochloride represents pharmaceutical-grade excellence with a guaranteed assay of 98.5-101.0% and impurity levels maintained at ≤0.1%, ensuring superior quality for anti-diabetic medication production. This active pharmaceutical ingredient is manufactured under strict quality control protocols, making it the preferred choice for companies seeking reliable raw materials for diabetes treatment formulations. The product's consistent purity and performance characteristics make it suitable for both immediate-release and extended-release drug delivery systems, providing formulators with versatile options for medication development and manufacturing. The pharmaceutical industry extensively utilizes Metformin Hydrochloride in manufacturing anti-diabetic medications, particularly for type 2 diabetes management. Pharmaceutical companies, contract manufacturing organizations, and generic drug manufacturers rely on this high-quality API for producing tablets, combination drugs, and various oral formulations. The compound's established safety profile and therapeutic efficacy make it fundamental to diabetes treatment protocols worldwide, serving both prescription and medication markets across different healthcare systems and regulatory environments while meeting international pharmacopeia standards. Businesses choosing this Metformin Hydrochloride benefit from exceptional reliability, consistent quality, and OEM manufacturing options that meet specific formulation requirements. The product's stringent quality control measures ensure batch-to-batch consistency, reducing production risks and compliance issues. With negotiable pricing structures and guaranteed on-time delivery, this API offers manufacturers competitive advantages in production planning, cost management, and market responsiveness, ultimately supporting better patient outcomes through reliable medication supply chains and consistent therapeutic performance. Key Features: - Pharmaceutical-grade Metformin Hydrochloride with 98.5-101.0% assay purity - Maximum impurity level of ≤0.1% ensuring medication safety and efficacy - Ignition residue maintained at ≤0.1% for superior quality standards - OEM supply options available for manufacturing requirements - Chemical identity: 1,1-Dimethylbiguanide monohydrochloride as specified Benefits: - Consistent high purity ensures reliable therapeutic performance in medications - Low impurity levels reduce production risks and compliance issues - Strict quality control guarantees batch-to-batch consistency and reliability - OEM manufacturing support enables customized formulation development - Proven safety profile supports diverse pharmaceutical applications
Specifications
| Attribute | Value |
|---|---|
| Type | Metformin Hydrochloride |
| Impurity | ≤0.1% |
| Ignition | ≤0.1% |
| Supply Type | OEM |
| Price | Negotiable |
| Delivery | On Time Delivery |
| Application | Industrial Applications |
| Assay | 98.5-101.0% |
| Therapeutic Usage | Anti-Diabetic |
| Chemical Name | 1,1-Dimethylbiguanide monohydrochloride |
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Metformin Hydrochloride serves as the primary active ingredient in anti-diabetic medications, specifically formulated for type 2 diabetes management. Its high purity and consistent assay make it ideal for tablet formulations, extended-release medications, and combination therapies requiring precise dosing and reliable therapeutic outcomes. This pharmaceutical-grade ingredient meets stringent quality standards, ensuring safety and efficacy in finished products for healthcare markets worldwide. The compound's versatility extends to various pharmaceutical applications beyond standard diabetes treatment. Formulation scientists utilize this API for developing innovative drug delivery systems and combination therapies. Its established safety profile and compatibility with multiple excipients make it suitable for specialized formulations including pediatric medications and geriatric treatments that demand careful dosage management and minimal side effects. - Manufacturing immediate-release tablets for type 2 diabetes treatment - Producing extended-release metformin formulations for improved patient compliance - Developing combination drugs incorporating metformin with other anti-diabetic agents - Creating pediatric diabetic medications with precise dosing requirements - Formulating generic pharmaceutical products for accessible diabetes treatment
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Kothari Phyto Chemicals International, Tamil Nadu
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory Size1000-5000 sqm
Annual Production Capacity0
Main Markets
Eastern Asia
OEM Metformin Hydrochloride Anti Diabetic API 98.5 101.0% Assay
OEM Metformin Hydrochloride anti-diabetic API with 98.5-101.0% assay purity and ≤0.1% impurities for pharmaceutical manufacturers requiring reliable diabetes medication production.
Min. Order Quantity: 1 Tons
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Kothari Phyto Chemicals International
Health & Personal CareManufacturer