Apparel & Fashion
Loading subcategories...
View More
Description
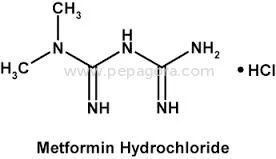
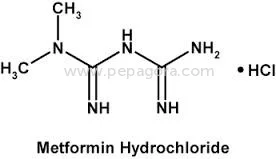
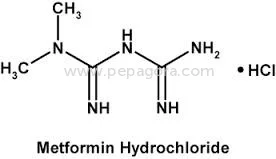
High Purity Metformin Hydrochloride API represents pharmaceutical-grade excellence with a precise assay range of 98.5-101.0%, specifically designed for anti-diabetic medication for...
High Purity Metformin Hydrochloride API represents pharmaceutical-grade excellence with a precise assay range of 98.5-101.0%, specifically designed for anti-diabetic medication formulations. This active pharmaceutical ingredient maintains impurity levels at ≤0.1% and ignition residue controlled at ≤0.1%, meeting stringent pharmacopeial standards including USP and EP requirements. The chemical compound, known scientifically as 1,1-Dimethylbiguanide monohydrochloride, demonstrates exceptional stability and compatibility with various pharmaceutical excipients. Our manufacturing process incorporates rigorous quality control measures at every stage, ensuring consistent purity and reliability that pharmaceutical manufacturers can trust for their critical formulations. The OEM supply model provides manufacturing flexibility while maintaining the highest quality standards required for medical applications. This pharmaceutical-grade Metformin Hydrochloride serves the global healthcare industry, particularly manufacturers producing medications for Type 2 diabetes management. Pharmaceutical companies worldwide utilize this API in creating both generic and branded anti-diabetic drugs, including tablets, capsules, and combination therapies. The product meets the requirements of manufacturers complying with international regulatory standards from agencies including FDA and EMA. Industries benefit from the API's versatility across various production scales, from small batch formulations to large-scale manufacturing operations. The consistent quality supports pharmaceutical companies in maintaining their product efficacy and safety profiles while meeting global market demands for diabetes treatment solutions. The business value of this Metformin Hydrochloride API lies in its unwavering reliability and consistent quality that pharmaceutical manufacturers can depend on for uninterrupted production cycles. Our established supply chain ensures timely delivery commitments, helping manufacturers maintain their production schedules and market supply obligations. The competitive pricing structure provides cost-effectiveness for volume purchases without compromising the stringent quality standards required for pharmaceutical ingredients. The controlled impurity profile and strict quality parameters demonstrate our commitment to exceeding industry standards, reducing batch rejection risks and ensuring consistent therapeutic performance. Manufacturers can confidently integrate this API into their formulations, knowing they receive a product that meets the highest pharmaceutical standards. Key Features: - High purity Metformin Hydrochloride with precise 98.5-101.0% assay range - Controlled impurity levels at ≤0.1% ensuring pharmaceutical-grade quality - Strict ignition residue control maintained at ≤0.1% for enhanced safety - Chemical identity: 1,1-Dimethylbiguanide monohydrochloride - OEM supply model supporting flexible manufacturing requirements Benefits: - Consistent therapeutic performance in anti-diabetic medication formulations - Reduced batch rejection risks through stringent quality control measures - Reliable supply chain ensuring uninterrupted manufacturing operations - Compliance with international pharmacopeial standards and regulations - Cost-effective solution for large-scale pharmaceutical production needs
Specifications
| Attribute | Value |
|---|---|
| Type | Metformin Hydrochloride |
| Impurity | ≤0.1% |
| Ignition | ≤0.1% |
| Supply Type | OEM |
| Price | Negotiable |
| Delivery | On Time Delivery |
| Application | Industrial Applications |
| Assay | 98.5-101.0% |
| Therapeutic Usage | Anti-Diabetic |
| Chemical Name | 1,1-Dimethylbiguanide monohydrochloride |
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Metformin Hydrochloride API serves as the primary active ingredient in anti-diabetic medications, effectively managing blood glucose levels in Type 2 diabetes patients. Its mechanism reduces hepatic glucose production while improving insulin sensitivity, making it essential for diabetes treatment protocols worldwide. The high-purity formulation ensures consistent therapeutic outcomes and patient safety across various medication formats and delivery systems. Pharmaceutical manufacturers utilize this API in multiple dosage forms including immediate-release tablets, extended-release formulations, and combination therapies. The consistent quality and reliable supply chain support large-scale production requirements while maintaining compliance with international pharmacopeial standards. Our manufacturing processes ensure batch-to-batch consistency that pharmaceutical companies can depend on for their production schedules and quality assurance protocols. - Core active ingredient in immediate-release metformin tablets for diabetes management - Primary component in extended-release formulations for improved patient compliance - Essential raw material for combination drugs with other anti-diabetic agents - Critical API for pharmaceutical companies manufacturing diabetes medications - Key ingredient for generic drug manufacturers supplying global healthcare markets
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Kothari Phyto Chemicals International, Tamil Nadu
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory Size1000-5000 sqm
Annual Production Capacity0
Main Markets
Eastern Asia
High Purity Metformin Hydrochloride API 98.5 101.0% Assay Anti Diabetic
High Purity Metformin Hydrochloride API 98.5-101.0% Assay for anti-diabetic medications. Pharmaceutical-grade active ingredient with ≤0.1% impurities, reliable OEM supply for diabetes treatment formulations.
Min. Order Quantity: 1 Tons
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Kothari Phyto Chemicals International
Health & Personal CareManufacturer