Apparel & Fashion
Loading subcategories...
View More
Description
High Purity Metformin Hydrochloride API represents a critical pharmaceutical ingredient for diabetes treatment worldwide. With a precise assay range of 98.5-101.0% and impurities s...
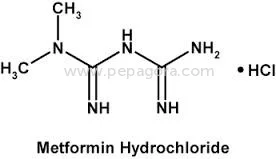
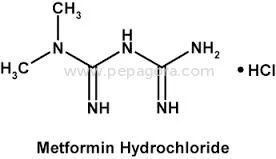
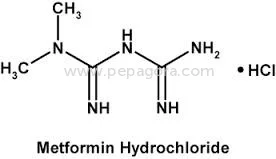
High Purity Metformin Hydrochloride API represents a critical pharmaceutical ingredient for diabetes treatment worldwide. With a precise assay range of 98.5-101.0% and impurities strictly controlled at ≤0.1%, this active pharmaceutical ingredient meets rigorous pharmacopeial standards for quality, safety, and efficacy. Chemically identified as 1,1-Dimethylbiguanide monohydrochloride, this compound offers excellent stability and predictable pharmacokinetic properties essential for reliable medication manufacturing. Our OEM supply model provides pharmaceutical companies with consistent access to this vital diabetes treatment component, supported by comprehensive quality assurance protocols and dependable delivery schedules that ensure uninterrupted production cycles. The pharmaceutical industry relies on Metformin Hydrochloride API as the foundation for both generic and branded anti-diabetic medications. Manufacturers incorporate this high-quality API into various therapeutic formulations including standard immediate-release tablets, controlled-release versions for prolonged effect, and innovative combination products with complementary anti-diabetic agents. The compound's well-documented safety profile and proven clinical efficacy have established it as a preferred first-line treatment recommended by global health authorities. Pharmaceutical producers appreciate its seamless integration with standard manufacturing equipment and common excipients, enabling efficient scale-up from development to full commercial production while maintaining batch-to-batch consistency and regulatory compliance. Businesses selecting our Metformin Hydrochloride API gain substantial operational advantages through reliable supply partnerships and uncompromising quality consistency. The stringent impurity control at ≤0.1% and tight assay specifications ensure finished medications satisfy regulatory requirements across multiple international markets, minimizing compliance risks and accelerating market entry timelines. Our OEM capabilities support pharmaceutical companies in developing differentiated products while maintaining cost efficiency throughout the manufacturing process. The proven reliability of this API translates directly to reduced production variability, lower batch rejection rates, and ultimately, trustworthy diabetes medications that healthcare professionals prescribe with confidence and patients depend on for effective long-term disease management. Key Features: - High purity Metformin Hydrochloride API with 98.5-101.0% assay meeting pharmacopeial standards - Extremely low impurity profile at ≤0.1% ensuring medication safety and efficacy - ≤0.1% ignition residue for superior product quality and manufacturing consistency - OEM supply model with reliable delivery schedules for uninterrupted production - Chemical identity: 1,1-Dimethylbiguanide monohydrochloride with established stability Benefits: - Consistent quality that meets international regulatory standards for pharmaceutical manufacturing - Reduced compliance risks through stringent impurity control and precise assay specifications - Reliable supply chain with OEM flexibility supporting diverse production requirements - Enhanced manufacturing efficiency with lower batch variability and rejection rates - Trusted ingredient foundation for effective diabetes medications prescribed globally
Specifications
| Attribute | Value |
|---|---|
| Type | Metformin Hydrochloride |
| Impurity | ≤0.1% |
| Ignition | ≤0.1% |
| Supply Type | OEM |
| Price | Negotiable |
| Delivery | On Time Delivery |
| Application | Industrial Applications |
| Assay | 98.5-101.0% |
| Therapeutic Usage | Anti-Diabetic |
| Chemical Name | 1,1-Dimethylbiguanide monohydrochloride |
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Metformin Hydrochloride API serves as the essential active ingredient in oral medications for managing Type 2 diabetes. Its mechanism reduces liver glucose production and enhances insulin sensitivity, making it a globally recognized first-line treatment. This high-purity API enables manufacturers to produce consistent, effective diabetes therapies with reliable therapeutic outcomes that meet international quality standards. Pharmaceutical manufacturers value this API for its excellent stability and compatibility with standard production processes and excipients. This allows for flexible formulation across various dosage forms including immediate-release tablets, extended-release versions, and combination therapies. The consistent quality supports scalable production from pilot batches to full commercial manufacturing, ensuring medications meet diverse patient needs and regulatory requirements across different markets. - Manufacturing immediate-release metformin tablets for effective blood sugar management - Producing extended-release formulations that allow convenient once-daily dosing - Creating combination medications with other anti-diabetic agents like sitagliptin or glimepiride - Bulk production for government healthcare programs and public health initiatives - Manufacturing export-quality diabetes medications for international pharmaceutical markets
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Kothari Phyto Chemicals International, Tamil Nadu
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory Size1000-5000 sqm
Annual Production Capacity0
Main Markets
Eastern Asia
High Purity Metformin Hydrochloride API 98.5 101.0% Assay Anti Diabetic
High Purity Metformin Hydrochloride API with 98.5-101.0% assay and ≤0.1% impurities for reliable anti-diabetic medication manufacturing. Trusted OEM supply for pharmaceutical production globally.
Min. Order Quantity: 1 Tons
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Kothari Phyto Chemicals International
Health & Personal CareManufacturer