Apparel & Fashion
Loading subcategories...
View More
Description
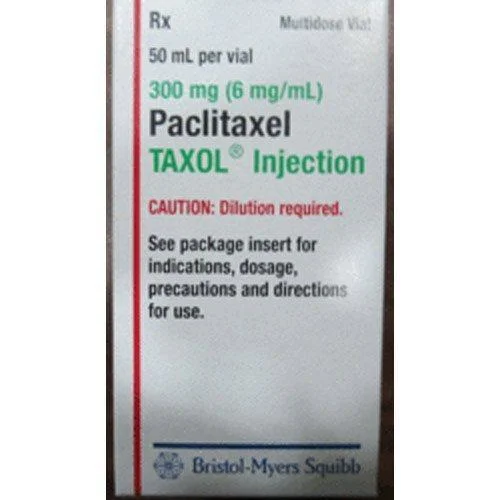
Inj Taxol provides healthcare providers with a trusted chemotherapy solution containing paclitaxel, an established antineoplastic agent that stabilizes cellular microtubules to dis...
Inj Taxol provides healthcare providers with a trusted chemotherapy solution containing paclitaxel, an established antineoplastic agent that stabilizes cellular microtubules to disrupt cancer cell division. Manufactured under rigorous current Good Manufacturing Practice (cGMP) standards, each batch undergoes comprehensive quality verification for potency, purity, and sterility. This injectable formulation offers predictable pharmacokinetics and a well-characterized safety profile, making it a reliable component in cancer treatment regimens. The product's consistent performance and quality assurance protocols give medical teams confidence in its therapeutic effects, supporting standardized treatment approaches across diverse healthcare settings. This chemotherapy agent serves oncology departments in tertiary hospitals, dedicated cancer centers, and outpatient infusion clinics worldwide. It is routinely employed in treating breast carcinoma, epithelial ovarian cancer, non-small cell lung cancer, and Kaposi's sarcoma. The drug fits into multidisciplinary cancer management, often used alongside surgical interventions, radiation therapy, and newer targeted treatments. Procurement specialists stock it for hospital pharmacies and chemotherapy preparation units that serve both public health systems and private oncology practices, ensuring availability for scheduled treatment cycles. Healthcare institutions value Inj Taxol for its therapeutic reliability and supply chain consistency, which directly supports patient care continuity. The manufacturing process emphasizes batch-to-batch uniformity, reducing variability in clinical response. Medical directors appreciate its established efficacy data and compatibility with various infusion protocols, allowing flexible integration into existing workflows. The product's stability profile and validated shelf life facilitate efficient inventory management, minimizing waste while meeting treatment demand. Its availability in multiple vial sizes helps facilities optimize procurement based on patient volume and dosing requirements. Key Features: - Contains paclitaxel as the active pharmaceutical ingredient with high purity specifications - Produced in cGMP-certified facilities with documented quality systems - Available in various dosage strengths and vial configurations - Undergoes sterility testing, endotoxin analysis, and potency verification - Formulated for stability with clearly defined storage conditions Benefits: - Delivers consistent therapeutic effect through standardized manufacturing - Supports treatment protocol adherence with reliable quality and potency - Enables flexible inventory management with multiple packaging options - Reduces clinical uncertainty through batch-to-batch consistency - Integrates smoothly into existing chemotherapy preparation workflows
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Inj Taxol is administered by oncology professionals in hospital infusion centers and specialized cancer clinics. It is a cornerstone of systemic chemotherapy protocols, delivered via controlled intravenous infusion to ensure precise dosing and patient safety during treatment cycles. This injectable formulation integrates seamlessly into established cancer care pathways, supporting both inpatient and outpatient treatment settings with consistent therapeutic performance. Medical oncologists utilize Inj Taxol across multiple cancer types, incorporating it into neoadjuvant, adjuvant, and palliative treatment strategies. The drug's mechanism targets rapidly dividing cells, making it effective against various solid tumors when used according to evidence-based guidelines. Treatment protocols typically combine it with other agents or employ it as monotherapy based on cancer staging, patient tolerance, and therapeutic objectives determined by the clinical team. - Standard chemotherapy for metastatic breast cancer, often combined with other agents - First-line treatment for advanced ovarian cancer following cytoreductive surgery - Therapeutic option for non-small cell lung cancer in specific patient profiles - Management of AIDS-related Kaposi's sarcoma in comprehensive HIV care - Adjuvant treatment for node-positive breast cancer to reduce recurrence risk
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Aark Pharmaceuticals, New Delhi
Trading CompanyDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Inj Taxol Chemotherapy Drug For Cancer Treatment
Inj Taxol chemotherapy drug treats breast, ovarian, and lung cancers by inhibiting cancer cell division. This reliable, high-purity paclitaxel injection is manufactured under strict cGMP standards for global hospitals and oncology centers.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Aark Pharmaceuticals
Trading Company