Apparel & Fashion
Loading subcategories...
View More
Description
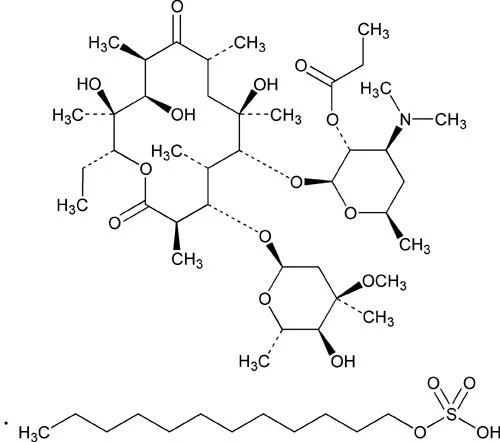
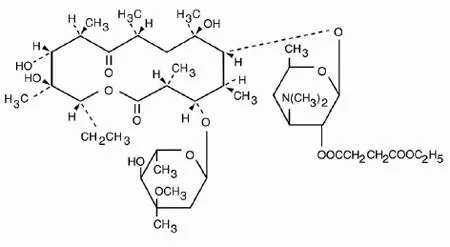
Erythromycin Propionate FP represents a high-purity macrolide antibiotic salt specifically engineered for pharmaceutical manufacturing applications. Derived from Streptomyces eryth...
Erythromycin Propionate FP represents a high-purity macrolide antibiotic salt specifically engineered for pharmaceutical manufacturing applications. Derived from Streptomyces erythreus, this specialized compound functions as the lauryl sulfate salt of erythromycin's propionic ester, offering enhanced stability and consistent performance in various formulations. Its primary mechanism involves bacteriostatic action through binding to the 50S ribosomal subunits in susceptible bacteria, effectively inhibiting peptidyl transferase activity and disrupting protein synthesis during translation. This targeted approach ensures reliable action against bacterial infections while maintaining the compound's integrity throughout the manufacturing process and shelf life. The high-purity nature of this API guarantees consistent results for pharmaceutical companies seeking dependable active ingredients for their anti-infective product lines. This antibiotic API finds extensive application across multiple therapeutic areas within the pharmaceutical industry. It serves as a fundamental component in medications addressing respiratory conditions including pneumonia, bronchitis, and whooping cough, where its bacteriostatic properties prove particularly effective. The dermatological sector incorporates it into treatments for skin infections such as acne, impetigo, and cellulitis, leveraging its ability to combat Gram-positive bacteria. Veterinary medicine utilizes this compound in antibiotics for livestock, poultry, and companion animals, supporting animal health management and disease prevention. Its compatibility with various formulation types—from oral suspensions and tablets to topical applications—makes it a versatile choice for manufacturers developing comprehensive anti-infective solutions across different treatment modalities and patient needs. Manufacturers benefit from the exceptional reliability and formulation suitability of Erythromycin Propionate FP, produced under rigorous quality control standards to ensure batch-to-batch consistency. The product's predictable bacteriostatic performance reduces development uncertainties and accelerates regulatory approval processes, enabling faster market entry for finished pharmaceutical products. Its chemical stability and compatibility with various excipients allow seamless integration into existing manufacturing processes while maintaining therapeutic efficacy. This reliability translates into strengthened product portfolios for pharmaceutical companies, enhanced treatment outcomes for end-users, and increased trust among healthcare providers prescribing medications containing this established antibiotic compound. The consistent quality and performance characteristics make it a valuable strategic component for businesses aiming to deliver effective anti-infective solutions. Key Features: - High-purity macrolide antibiotic salt derived from Streptomyces erythreus - Lauryl sulfate salt form of erythromycin propionic ester for enhanced stability - Bacteriostatic mechanism effectively inhibits bacterial protein synthesis - Specific binding to 50S ribosomal subunits disrupts peptidyl transferase activity - Consistent quality and performance across manufacturing batches Benefits: - Reliable bacteriostatic action against susceptible bacterial strains - Enhanced formulation stability for extended product shelf life - Consistent batch-to-batch quality ensuring predictable therapeutic outcomes - Compatibility with various drug delivery systems and formulation types - Accelerated development timelines for anti-infective pharmaceutical products
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Erythromycin Propionate FP serves as a crucial active ingredient in pharmaceutical formulations requiring precise bacteriostatic action. Its mechanism involves binding to bacterial ribosomes to inhibit protein synthesis, making it effective against various susceptible organisms. This high-purity antibiotic salt ensures consistent performance in multiple drug delivery systems while maintaining therapeutic efficacy across different treatment protocols. This specialized API is essential for developing oral, topical, and systemic medications targeting bacterial infections. Pharmaceutical manufacturers rely on its consistent quality for producing formulations that meet strict regulatory standards and therapeutic requirements. The product's stability and compatibility make it suitable for various pharmaceutical applications, from human medicines to veterinary treatments, ensuring reliable outcomes across different medical scenarios. - Manufacturing oral antibiotic suspensions for pediatric and adult respiratory infections - Formulating topical creams and ointments for bacterial skin condition treatments - Producing tablet medications for respiratory tract infection management - Developing veterinary antibiotics for livestock and companion animal health - Creating combination therapies for enhanced antibacterial treatment protocols
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mehta Pharmaceutical Industries, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical API Erythromycin Propionate FP High Purity Antibiotic
Pharmaceutical API Erythromycin Propionate FP high-purity macrolide antibiotic salt for manufacturing anti-infective medications, offering reliable bacteriostatic action and consistent quality for respiratory and skin infection treatments.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mehta Pharmaceutical Industries
Manufacturer