Apparel & Fashion
Loading subcategories...
View More
Description
Erythromycin Ethyl Succinate BP/USP is a high-purity antibiotic ester specifically designed for optimal oral administration, offering enhanced bioavailability and effective inhibit...
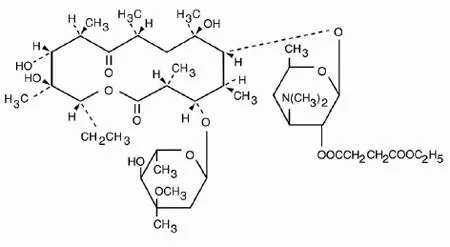
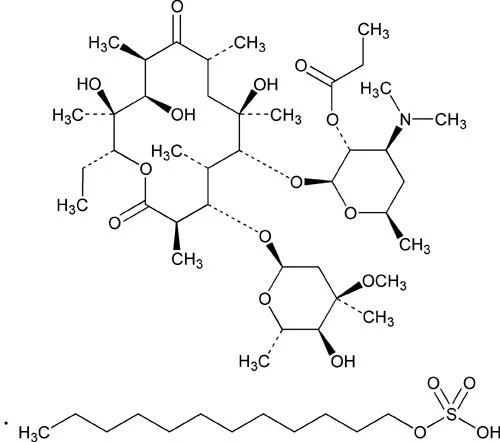
Erythromycin Ethyl Succinate BP/USP is a high-purity antibiotic ester specifically designed for optimal oral administration, offering enhanced bioavailability and effective inhibition of bacterial protein synthesis. Complying with BP and USP standards, this macrolide antibiotic ensures consistent quality and reliability for pharmaceutical applications. It demonstrates strong activity against Gram-positive bacteria and atypical pathogens such as Mycoplasma and Legionella, making it a versatile choice for treating various infections. With CAS number 41342-53-4, it delivers dependable performance in multiple formulations including tablets and suspensions, meeting rigorous global quality requirements. The pharmaceutical industry extensively employs Erythromycin Ethyl Succinate in manufacturing antibiotics for both human and veterinary medicine. It serves as a key ingredient in drugs targeting respiratory tract infections like pneumonia, bronchitis, and pertussis, as well as skin and soft tissue infections. Veterinary applications include combating respiratory diseases in livestock and pets, ensuring animal health and productivity. Clinical research sectors utilize this API for developing novel therapeutic combinations and studying antibiotic resistance mechanisms. Its adherence to international pharmacopeia standards makes it suitable for regulated markets worldwide, where quality and efficacy are essential for regulatory approval and commercial success. Businesses benefit from the proven reliability of Erythromycin Ethyl Succinate, which ensures consistent supply and formulation versatility for antibiotic products. Its established efficacy reduces development risks and accelerates market entry for new treatments. The API's broad-spectrum activity enables companies to address multiple infection types with a single ingredient, enhancing product portfolio diversity. Quality assurance through BP/USP compliance builds trust with regulators and healthcare providers, minimizing compliance challenges. Long-standing medical use underscores its safety profile and effectiveness, providing a competitive advantage in markets demanding dependable pharmaceutical ingredients. Key Features: - Complies with BP and USP standards for guaranteed purity and quality. - Effective against Gram-positive bacteria and atypical pathogens like Mycoplasma. - Suitable for oral administration with enhanced bioavailability. - Versatile for use in tablets, suspensions, and pediatric formulations. - Stable compound with consistent performance across various dosage forms. Benefits: - Accelerates product development with proven efficacy and safety profile. - Supports diverse therapeutic applications across human and veterinary medicine. - Enhances treatment outcomes with reliable bacterial protein synthesis inhibition. - Meets global regulatory requirements through pharmacopeia compliance. - Reduces manufacturing risks with high-purity, consistent quality API.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Erythromycin Ethyl Succinate BP/USP serves as a critical active pharmaceutical ingredient in oral antibiotic formulations, valued for its efficacy against respiratory and soft tissue infections. Its enhanced stability and bioavailability make it ideal for human and veterinary medicines, ensuring consistent performance in diverse therapeutic applications across global markets. This API is widely utilized in pharmaceutical manufacturing for developing tablets, suspensions, and pediatric formulations. Its broad-spectrum activity against Gram-positive bacteria and atypical pathogens supports treatments for community-acquired pneumonia, skin infections, and streptococcal pharyngitis, meeting stringent pharmacopeia standards for quality and safety. - Manufacturing oral antibiotic tablets for treating community-acquired pneumonia and bronchitis. - Formulating pediatric suspensions to combat streptococcal pharyngitis and skin infections. - Producing veterinary antibiotics for respiratory diseases in livestock and pets. - Developing combination therapies for enhanced efficacy against resistant bacterial strains. - Creating medications for sinusitis and otitis media in both prescription and OTC formats.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mehta Pharmaceutical Industries, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical API Erythromycin Ethyl Succinate BP/USP Antibiotic
Pharmaceutical API Erythromycin Ethyl Succinate BP/USP is a high-purity antibiotic ester for oral administration, effectively treating respiratory infections with reliable bacterial protein synthesis inhibition.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mehta Pharmaceutical Industries
Manufacturer