Apparel & Fashion
Loading subcategories...
View More
Description
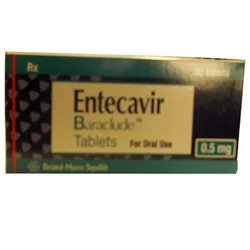
Entecavir Baraclude tablets represent a critical advancement in antiviral therapy for chronic hepatitis B infection. As a potent guanosine nucleoside analogue, Entecavir effectivel...
Entecavir Baraclude tablets represent a critical advancement in antiviral therapy for chronic hepatitis B infection. As a potent guanosine nucleoside analogue, Entecavir effectively suppresses HBV replication by inhibiting viral reverse transcriptase activity. Our supply provides pharmaceutical-grade tablets that meet stringent international quality standards, ensuring consistent dosage, bioavailability, and optimal patient outcomes. We deliver reliable access to this essential medication for healthcare distributors and institutions, with dependable logistics and support for various quantity requirements. This medication serves vital roles across multiple healthcare sectors, including large hospital networks, government health departments, specialized treatment centers, and pharmacy distribution chains. Hepatologists and infectious disease specialists utilize Entecavir as a first-line therapy for chronic HBV patients demonstrating active viral replication and liver inflammation. Its value is particularly significant in regions with high hepatitis B prevalence, where consistent access to quality antiviral medication directly improves public health outcomes and reduces long-term complications like liver failure. Our Entecavir Baraclude tablets offer exceptional value through reliable supply chain partnerships and unwavering product quality. We maintain strict protocols for storage, handling, and documentation to ensure product integrity from point of origin to final delivery. The proven high barrier to resistance associated with Entecavir ensures long-term treatment efficacy, which supports predictable patient outcomes and stable inventory planning for healthcare providers and distributors alike. Key Features: - Pharmaceutical-grade Entecavir manufactured to meet international pharmacopeial standards. - Available in multiple, precise dosage strengths for tailored treatment protocols. - Produced in WHO-GMP certified facilities guaranteeing batch-to-batch consistency. - Supported by comprehensive documentation including certificates of analysis. - Supplied with reliable, traceable logistics ensuring timely delivery. Benefits: - Provides a high-potency antiviral with a strong genetic barrier to resistance for effective long-term management. - Enables flexible treatment planning for healthcare providers through available dosage options. - Assures quality and safety for patient use via certified manufacturing and rigorous testing. - Simplifies procurement and compliance with complete, verifiable product documentation. - Supports uninterrupted patient care through dependable, consistent supply to distributors and institutions.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Entecavir Baraclude tablets are a potent nucleoside analogue antiviral essential for managing chronic hepatitis B virus infections in adults and pediatric patients. It effectively inhibits viral replication, reducing viral load and liver damage, and is prescribed for patients with compensated liver disease and evidence of active viral replication. Healthcare providers choose Entecavir for its high barrier to resistance compared to other treatments. It is suitable for both treatment-naïve patients and those with lamivudine resistance, helping prevent disease progression to cirrhosis and hepatocellular carcinoma, making it a cornerstone of long-term HBV management strategies. - Hospital pharmacies stock Entecavir for inpatient and outpatient hepatitis B treatment programs. - Government public health initiatives distribute these tablets through national healthcare programs. - Specialty liver and infectious disease clinics use it for patients with lamivudine-resistant hepatitis B strains. - Transplant centers administer Entecavir to prevent HBV recurrence following liver transplantation. - Retail pharmacy chains supply it as maintenance therapy for chronic hepatitis B patients in the community.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Biogenix Pharma UK Limited, Maharashtra
Manufacturer
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Supply Of Entecavir Baraclude Tablets For Hepatitis B Treatment
Supply Entecavir Baraclude tablets for hepatitis B treatment. Our pharmaceutical-grade antiviral medication offers reliable, timely delivery for distributors and healthcare providers worldwide.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Biogenix Pharma UK Limited
Manufacturer