Apparel & Fashion
Loading subcategories...
View More
Description
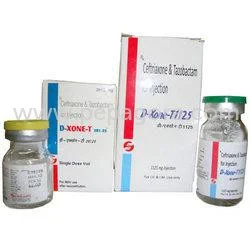
D XONE T Ceftriaxone Tazobactum Sterile Injection represents a sophisticated antibiotic solution combining Ceftriaxone Sodium IP 250mg with Tazobactum Sodium USP 31.25mg in a steri...
D XONE T Ceftriaxone Tazobactum Sterile Injection represents a sophisticated antibiotic solution combining Ceftriaxone Sodium IP 250mg with Tazobactum Sodium USP 31.25mg in a sterile dry formulation. This powerful combination leverages the broad-spectrum activity of third-generation cephalosporin with beta-lactamase inhibition, creating an effective therapeutic option against resistant bacterial strains. Manufactured under stringent pharmaceutical standards, each vial maintains consistent potency and purity from batch to batch. The dry powder format ensures extended stability and shelf life, requiring simple reconstitution with sterile water before intravenous or intramuscular administration by qualified healthcare professionals. This injection serves critical roles across multiple healthcare sectors including hospital pharmacies, intensive care units, surgical centers, and emergency medical services. Medical professionals deploy it for treating severe community-acquired and hospital-acquired infections across internal medicine, surgery, pediatrics, and critical care specialties. The product meets the rigorous demands of modern healthcare facilities confronting antibiotic resistance, providing reliable coverage where infection control directly impacts patient recovery and treatment outcomes. Its versatility makes it valuable in both inpatient and outpatient settings where serious bacterial infections require immediate, effective intervention. Procurement specialists and medical distributors value D XONE T for its proven therapeutic reliability and consistent quality standards. The manufacturing process adheres to strict quality control measures that guarantee product integrity and compliance with international pharmaceutical regulations. This dual-action antibiotic delivers clinical advantages that translate to improved patient outcomes, reduced treatment duration, and optimized healthcare resource utilization. The product's reliability and effectiveness make it an essential component of institutional formularies and medical supply chains focused on delivering high-quality patient care. Key Features: - Sterile dry injection containing Ceftriaxone Sodium IP equivalent to 250mg Ceftriaxone - Combined with Tazobactum Sodium USP equivalent to 31.25mg Tazobactum - Manufactured under strict sterile pharmaceutical conditions - Dry powder formulation ensuring stability and extended shelf life - Suitable for both intravenous and intramuscular administration Benefits: - Broad-spectrum antibiotic coverage against resistant bacterial strains - Beta-lactamase inhibition enhances effectiveness against resistant organisms - Consistent quality and reliable performance across all batches - Extended shelf life maintains potency throughout product lifecycle - Versatile administration options suit various clinical settings
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
D XONE T sterile injection provides critical antibiotic therapy for severe bacterial infections requiring immediate medical intervention. Each vial contains precisely measured Ceftriaxone Sodium IP 250mg and Tazobactum Sodium USP 31.25mg, ensuring accurate dosing for complex infections where treatment precision matters most. The combination delivers powerful antibacterial action against resistant strains while maintaining safety profiles suitable for various patient populations under professional supervision. This antibiotic formulation serves diverse medical settings including hospital wards, emergency departments, and specialized care units. Healthcare facilities rely on its consistent performance for managing complicated infections across multiple specialties. The product's stability and reliability make it essential inventory for pharmacies and medical centers that prioritize effective infection control protocols and positive patient outcomes through evidence-based antibiotic therapy. - Treatment of hospital-acquired pneumonia and severe respiratory infections - Management of complicated urinary tract infections requiring intravenous therapy - Surgical infection prevention and post-operative care protocols - Serious skin and soft tissue infections with suspected resistant organisms - Empirical treatment for febrile neutropenia in compromised patients
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Navdeep Bioceuticals, Haryana
Raw Materials & Chemicals•ManufacturerDistributor / Wholesaler
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
D XONE T Ceftriaxone Tazobactum Sterile Injection 250mg 31.25mg
D XONE T Ceftriaxone Tazobactum Sterile Injection 250mg 31.25mg delivers reliable antibiotic treatment for serious bacterial infections. This sterile dry formulation combines broad-spectrum coverage with beta-lactamase inhibition, trusted by healthcare providers worldwide for consistent quality and effectiveness.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Navdeep Bioceuticals
Raw Materials & ChemicalsManufacturer