Apparel & Fashion
Loading subcategories...
View More
Description
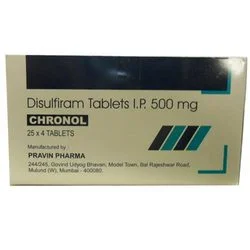
Disulfiram 500mg Tablets represent a specialized pharmaceutical intervention for alcohol dependence, manufactured to exacting USP standards for assured purity, potency, and safety....
Disulfiram 500mg Tablets represent a specialized pharmaceutical intervention for alcohol dependence, manufactured to exacting USP standards for assured purity, potency, and safety. Each white to off-white, round tablet is precisely formulated with 500mg of Disulfiram, the active ingredient that inhibits acetaldehyde dehydrogenase. This inhibition causes an accumulation of acetaldehyde when alcohol is consumed, resulting in highly unpleasant physical reactions that support aversion therapy. The tablets feature clear embossing with the letter 'A' on one side and a practical double score line on the other, enabling easy identification by healthcare staff and allowing for accurate dose splitting when clinically indicated. This commitment to precise manufacturing and thoughtful design underpins a medication trusted globally for its role in helping individuals achieve and sustain sobriety. This medication is utilized across a comprehensive spectrum of healthcare and institutional settings dedicated to treating alcohol use disorder. Hospital addiction medicine departments and psychiatric units incorporate it into integrated treatment plans. Private and non-profit rehabilitation centers rely on it for their residential programs, while outpatient clinics prescribe it for community-based recovery support. Public health agencies and government programs source these tablets for large-scale substance abuse initiatives. Furthermore, retail pharmacies stock them for prescriptions aimed at maintaining outpatient sobriety, and clinical research organizations may use them in studies advancing addiction treatment methodologies. Its application is fundamental wherever a reliable, evidence-based pharmacological support for abstinence is required. Procuring these Disulfiram tablets provides healthcare providers and distributors with a foundation of therapeutic reliability and supply chain confidence. The USP-grade certification is a direct indicator of stringent quality control, reducing procurement risk and ensuring consistent patient outcomes batch after batch. The distinctive tablet design minimizes medication errors, enhancing patient safety in busy clinical environments. Dependable manufacturing and a robust supply chain guarantee uninterrupted availability for critical treatment programs, preventing disruptions in patient care. This product embodies a partnership in quality, offering healthcare professionals a trusted tool that aligns with professional standards and supports their commitment to effective, long-term patient recovery. Key Features: - USP-grade Disulfiram 500mg ensures pharmaceutical-standard purity and potency - Distinctive embossing with 'A' and a double score line for secure identification and dosing flexibility - White to off-white round tablet format for clear visual verification - Manufactured under rigorous quality control and compliance protocols - Consistent formulation delivers reliable and predictable therapeutic action Benefits: - Supports effective alcohol aversion therapy, aiding long-term sobriety and recovery - USP quality standard provides assurance of safety, efficacy, and batch-to-batch consistency - Scored tablet design allows healthcare professionals to tailor dosage as needed - Reduces medication error risk through clear identification features - Reliable supply from trusted manufacturer ensures continuity for treatment programs
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Disulfiram 500mg tablets serve as a cornerstone in clinical alcohol aversion therapy, creating a deterrent effect that supports long-term sobriety. Their consistent USP-grade formulation ensures predictable therapeutic results, making them essential for structured treatment protocols where patient safety and medication reliability are non-negotiable. Healthcare providers depend on these tablets for their proven mechanism of action, which helps patients maintain abstinence by inducing unpleasant physical reactions upon alcohol consumption. These tablets are integral to both inpatient and outpatient care models, supplied to facilities managing chronic alcohol use disorder. The scored design facilitates precise dose titration by medical professionals, accommodating individual patient treatment plans. This versatility supports a wide range of healthcare delivery systems, from large institutional programs to community-based clinics, ensuring effective treatment is accessible across different care settings. Real-world applications: - Hospital inpatient detoxification and addiction treatment units - Residential rehabilitation centers for long-term recovery programs - Outpatient clinics and community mental health facilities - Government-funded public health initiatives for substance abuse - Private practice settings for ongoing sobriety maintenance
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Biogenix Pharma UK Limited, Maharashtra
Manufacturer
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Disulfiram 500mg Tablets for Alcohol Dependence Treatment USP Grade
Disulfiram 500mg Tablets USP Grade for alcohol dependence treatment. Premium quality aversion therapy medication trusted by hospitals, rehab centers, and pharmacies worldwide for reliable patient outcomes.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Biogenix Pharma UK Limited
Manufacturer