Apparel & Fashion
Loading subcategories...
View More
Description
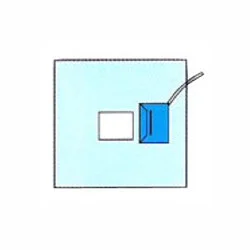
The Medical Craniotomy Drape is a specialized surgical barrier designed to meet the critical demands of neurosurgical procedures. It provides reliable protection against fluid stri...
The Medical Craniotomy Drape is a specialized surgical barrier designed to meet the critical demands of neurosurgical procedures. It provides reliable protection against fluid strike-through and microbial migration, helping to maintain a sterile operative field around the cranial site. Manufactured to comply with rigorous international medical standards, this drape is constructed from advanced materials that balance durability with the necessary drapability for complex anatomical contours. Its primary function is to act as a dependable first line of defense in infection prevention during some of medicine's most delicate operations. This drape is indispensable across the spectrum of neurosurgical care, from high-volume academic medical centers and specialized private neurosurgery hospitals to emergency trauma facilities. It is routinely used in procedures including craniotomies for tumor removal, surgery for cerebrovascular disorders like aneurysms, treatment of severe head trauma, and functional neurosurgery for conditions such as epilepsy. In these environments, the product supports surgical teams in adhering to the highest protocols for aseptic technique, which is fundamental to patient safety and positive clinical outcomes. Healthcare distributors and facilities investing in this craniotomy drape secure a product synonymous with reliability and performance. Its consistent quality helps reduce the risk of costly surgical site infections and associated complications, directly supporting both patient care standards and operational efficiency in the operating room. The drape's dependable construction ensures it performs as expected in critical moments, providing peace of mind to surgical teams and procurement managers alike. This focus on unwavering quality and suitability for its intended use makes it a trusted choice for serious medical suppliers and institutions. Key Features: - Engineered specifically for the fluid management and sterility challenges of cranial surgery - Constructed from advanced, fluid-resistant barrier materials for reliable protection - Designed for compatibility with various neurosurgical approaches and operating room setups - Manufactured in compliance with recognized international quality and safety standards - Provides a secure and maintainable sterile field around the surgical incision site Benefits: - Enhances infection control by significantly reducing the risk of intraoperative contamination - Improves procedural efficiency with effective fluid management and secure draping - Supports compliance with stringent hospital protocols for sterile technique - Delivers reliable performance that surgical teams can depend on for critical cases - Contributes to positive patient outcomes by helping to prevent surgical site infections
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
This premium craniotomy drape is engineered for the demanding environment of neurosurgery, where maintaining an absolute sterile field is non-negotiable. It offers exceptional fluid control to manage irrigation and bodily fluids during cranial access, significantly reducing the risk of contamination and surgical site infections. The design supports various surgical approaches and integrates seamlessly with modern operating theater equipment and protocols. Essential for advanced healthcare settings, this drape is a critical component in university hospitals, dedicated neurosurgical centers, and trauma units. Its reliability makes it suitable for both planned elective surgeries and urgent interventions, providing surgical teams with the confidence to maintain sterile conditions throughout lengthy and complex procedures. It is designed to meet stringent infection control standards and the specific draping challenges presented by cranial surgery. - Brain tumor resection and biopsy procedures requiring uncompromised sterility - Craniotomy for evacuation of hematomas or treatment of traumatic brain injury - Surgical interventions for epilepsy, including grid placement or resection - Cerebrovascular surgeries such as aneurysm clipping and AVM repairs - Complex skull base surgeries and cranial reconstructive procedures
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
D & U Health Age Private Limited, Gujarat
Health & Personal Care•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Medical Craniotomy Drape Barrier Protection Fluid Control
Medical Craniotomy Drape provides superior barrier protection and fluid control for neurosurgical procedures. Trusted by hospitals worldwide for quality and compliance with international standards.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
D & U Health Age Private Limited
Health & Personal CareManufacturer